- Title
-
Temporal-Spatial Resolution Fate Mapping Reveals Distinct Origins for Embryonic and Adult Microglia in Zebrafish
- Authors
- Xu, J., Zhu, L., He, S., Wu, Y., Jin, W., Yu, T., Qu, J.Y., Wen, Z.
- Source
- Full text @ Dev. Cell
|
The IR-LEGO-CreER-loxP System Provides a High Temporal-Spatial Resolution Labeling in Zebrafish RBI, VDA, and PBI Regions (A) Overexposed WISH staining shows that local 1,345 nm laser irradiation induces CreERT2 expression in the RBI, VDA, and PBI regions. Red arrows indicate the overexposed CreERT2 staining. (B) The distribution of the RBI-, VDA-region-, or PBI-derived cells (coro1a-GFP+ cells) in the brain, middle trunk, and tail at 4 dpf. The RBI-derived cells (top) were found in brain (dorsal view, rostral to the left, midbrain region), middle trunk, and tail (blue and orange boxed regions shown in Figure 1B), whereas the VDA-region-derived (middle) and PBI-derived (bottom) cells were detected in the trunk and tail but rarely in the brain. White arrows indicate GFP+ cells. (C) Quantification of GFP+ cells derived from the RBI (n = 9), VDA region (n = 10), or PBI (n = 6) in the brain. Error bars represent mean ± SEM. p < 0.001. (D) Quantification of GFP+ cells derived from the RBI (n = 6), VDA region (n = 6), or PBI (n = 6) in the middle trunk. Error bars represent mean ± SEM. p < 0.05. (E) Quantification of GFP+ cells derived from the RBI (n = 6), VDA region (n = 6), or PBI (n = 6) in the tail. Error bars represent mean ± SEM. p < 0.05; p < 0.001. See also Figure S1. |
|
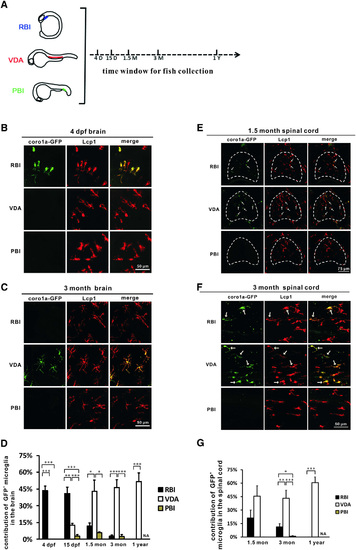
Embryonic/Larval and Adult Microglia in Zebrafish CNS Arise from Distinct Origins (A) A timeline shows the experimental setup. The RBI-, VDA-, and PBI-region-labeled fish were raised and collected at 4 dpf, 15 dpf, 1.5 months, 3 months, and 1 year for analysis. (B) Immunohistochemistry showed that the RBI- but not VDA- and PBI-region-derived cells (coro1a-GFP+ cells) constitute the major population of microglia (Lcp1+ cells) in the brains of 4-dpf zebrafish (dorsal view, rostral to the left, a part of the optic tectum). (C) Immunohistochemistry showed that the VDA- but not RBI- and PBI-derived cells (coro1a-GFP+ cells) constitute the major population of microglia (Lcp1+ cells) in the brains of 3-month-old adult zebrafish (sectioned brain). (D) Quantification of the contribution of the RBI-, VDA-, and PBI-region -derived microglia in the developing and adult zebrafish brain. For the RBI-labeled 4-dpf, 15-dpf, 1.5-month, 3-month, and 1-year adult fish, n = 8, 8, 5, 8, and 10, respectively; for the VDA-region-labeled 4-dpf, 15-dpf, 1.5-month, 3-month, and 1-year adult fish, n = 5, 6, 5, 8, and 9, respectively; for the PBI-labeled 4-dpf, 15-dpf, 1.5-month, and 3-month adult fish, n = 7, 6, 7, and 8 respectively. Error bars represent mean ± SEM. p < 0.05; p < 0.01; p < 0.001. (E) Immunohistochemistry showed the contribution of the RBI-, VDA-, and PBI-region-derived cells (coro1a-GFP+ cells) to microglia (Lcp1+ cells) in the sectioned 1.5-month zebrafish spinal cord (indicated by the dashed lines). White arrows indicate the coro1a-GFP+ microglia in the spinal cord. (F) Immunohistochemistry showed the contribution of the RBI-, VDA-, and PBI-region-derived cells (coro1a-GFP+ cells) to microglia (Lcp1+ cells) in the whole-mount 3-month adult zebrafish spinal cord. White arrows indicate the coro1a-GFP+ microglia in the spinal cord. (G) Quantification of the contribution of RBI-, VDA-, and PBI-region-derived microglia in 1.5-month, 3-month, and 1-year adult zebrafish spinal cord. For the RBI-labeled 1.5-month, 3-month, and 1-year adult fish, n = 6, 12, and 8, respectively; for the VDA-region-labeled 1.5-month, 3-month, and 1-year adult fish, n = 4, 5, and 9, respectively; for the PBI-labeled 1.5-month and 3-month adult fish, n = 5 and 6, respectively. Error bars represent mean ± SEM. p < 0.05; p < 0.01; p < 0.001. See also Figure S2. |
|
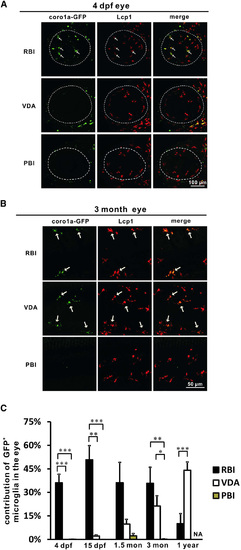
The Microglia in the Eye Exhibit Distinct Colonization and Composition Profile to the Microglia in the CNS (A) The RBI- but not VDA- and PBI-region-derived cells (coro1a-GFP+ cells) constitute the major population of microglia (Lcp1+ cells) in the eyes (indicated by dashed lines) of 4-dpf zebrafish. White arrows indicate coro1a-GFP+ microglia in the eyes. (B) The RBI- and VDA- but not PBI-region-derived cells (coro1a-GFP+ cells) constitute the major population of microglia (Lcp1+ cells) in the 3-month adult zebrafish eye. White arrows indicate the coro1a-GFP+ microglia in the eyes. (C) Quantification of the contribution of the RBI-, VDA-, and PBI-region-derived microglia in the developing and adult zebrafish eyes. For the RBI-labeled 4-dpf, 15-dpf, 1.5-month, 3-month, and 1-year adult fish, n = 8, 8, 5, 8, and 10, respectively; for the VDA-region-labeled 4-dpf, 15-dpf, 1.5-month, 3-month, and 1-year adult fish, n = 5, 6, 5, 8, and 9, respectively; for the PBI-labeled 4-dpf, 15-dpf, 1.5-month, and 3-month adult fish, n = 7, 6, 7, and 8, respectively. Error bars represent mean ± SEM. p < 0.05; p < 0.01; p < 0.001. |
|
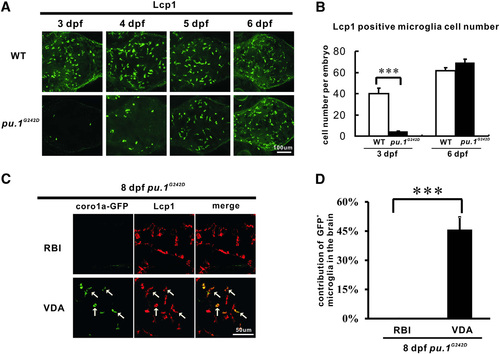
The RBI- but not VDA-Region-Derived Microglia Are Depleted in pu.1G242D Mutants (A) Microglia are largely absent in pu.1G242D mutants at 3 dpf, but are fully recovered at 6 dpf. Stacked confocal images of immunohistochemistry staining of Lcp1 of 3-dpf, 4-dpf, 5-dpf, and 6-dpf WT and pu.1G242D embryos from the dorsal view of the brain shows the recovery of microglia in pu.1G242D mutants from 5 dpf onward. (B) Quantification of Lcp1+ microglia number in WT (n = 6) and pu.1G242D embryos (n = 6) at 3 dpf and 6 dpf. Error bars represent mean ± SEM. p < 0.001. (C) Coro1a-GFP+ microglia are observed in VDA- but not RBI-region-labeled pu.1G242D embryos at 8 dpf. White arrows indicate the coro1a-GFP+ microglia in the brain (dorsal view, rostral to the left, a part of the optic tectum). (D) Quantification of the contribution of RBI- and VDA-region-derived microglia in 8dpf pu.1G242D embryos. For the RBI- and VDA-region-labeled embryos, n = 3 and 6, respectively. Error bars represent mean ± SEM. p < 0.001. |
|
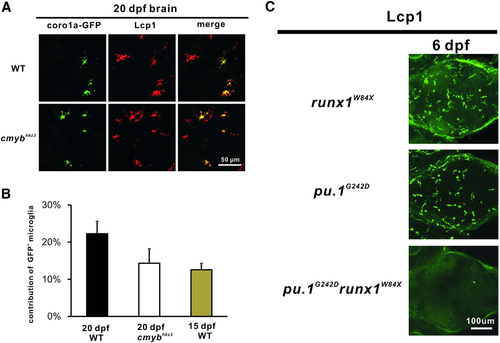
The Development of VDA Region-Derived Microglia Is Independent of cMyb but Requires Runx1 (A) The VDA-region-derived coro1a-GFP+ cells contribute to brain microglia in 20 dpf WT and cmybhkz3 mutant embryos (sectioned brain). (B) Quantification of the contribution of the VDA-region-derived brain microglia in 20-dpf WT (n = 8), cmybhkz3 mutant (n = 8), and 15-dpf WT larvae (n = 6). Error bars represent mean ± SEM. (C) Microglia fail to recover in the pu.1G242Drunx1W84X double mutants at 6 dpf. Stacked confocal images of immunohistochemistry staining of Lcp1 of 6 dpf runx1W84X, pu.1G242D single mutants, and pu.1G242Drunx1W84X double mutant from the dorsal view of the brain. See also Figure S3. |
|
The IR-LEGO-CreER-loxP System Induces Specific GFP Expression in the Embryos Treated with Infrared Irradiation and 4-OHT. (A) High magnification of a less exposed staining of CreER in situ hybridization in RBI, VDA and PBI regions. (B) Digitally reconstituted confocal images reveal GFP signals (indicated by arrows) in the embryos one day after heat shock in the RBI, VDA and PBI regions and 4-OHT treatment. |
|
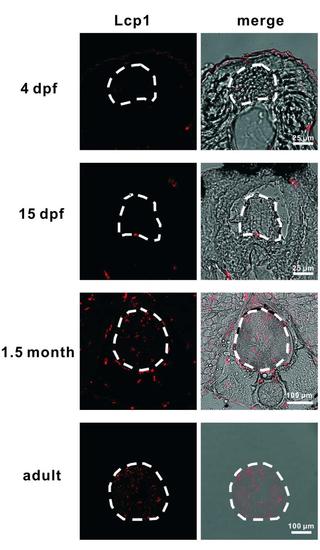
Microglia Colonize the Spinal Cord during Zebrafish Development. Lcp1+ microglia are detected in the spinal cord transverse sections of 1.5 month and adult fish but not of the 4 dpf and 15 dpf fish. Left panels are Lcp1+ signal and right panels represent merged views of Lcp1+ signal and bright field. The spinal cord is outlined by dashed lines. |
|
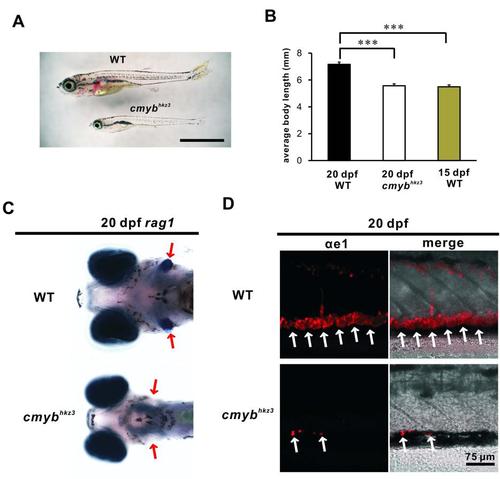
The cmybhkz3 Mutants Exhibit a Developmental Delay and Lack T Lymphocytes and Erythrocytes. (A) Imaging of 20 dpf WT and cmybhkz3 mutant. The upper larva is the WT and the lower larva is the cmybhkz3 mutant. The scale bar represents 2 mm. (B) Quantification of total body length of 20 dpf WT (n=11), cmybhkz3 mutant (n=10) and 15 dpf WT larvae (n=11). Error bars represent mean SEM. ***, P<0.001. (C) T cell marker rag1 is greatly reduced in 20 dpf cmybhkz3 mutants (lower panel) comparing with WT (upper panel). Red arrows indicate the position of thymus. (D) Only trace amount of erythrocyte marker hemoglobin αe1 is detected in the cmybhkz3 mutant (lower panel) comparing with WT (upper panel) at 20 dpf. White arrows indicate erythrocytes in the tail region. |
Reprinted from Developmental Cell, 34, Xu, J., Zhu, L., He, S., Wu, Y., Jin, W., Yu, T., Qu, J.Y., Wen, Z., Temporal-Spatial Resolution Fate Mapping Reveals Distinct Origins for Embryonic and Adult Microglia in Zebrafish, 632-641, Copyright (2015) with permission from Elsevier. Full text @ Dev. Cell