Fig. 4
- ID
- ZDB-FIG-221114-10
- Publication
- Wang et al., 2021 - atg7-Based Autophagy Activation Reverses Doxorubicin-Induced Cardiotoxicity
- Other Figures
- All Figure Page
- Back to All Figure Page
|
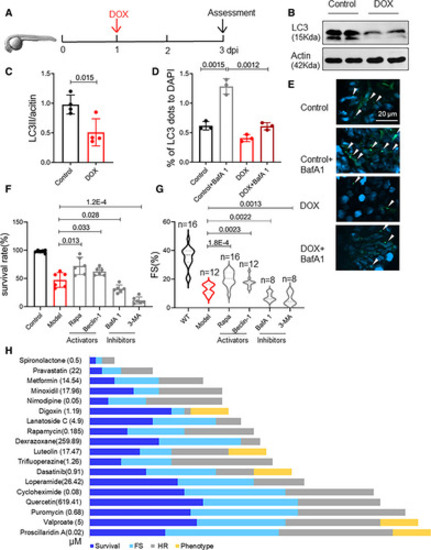
Food and Drug Administration–approved autophagy activators (FAAs) were ranked based on their therapeutic effects in a zebrafish embryonic anthracycline-induced cardiotoxicity (eAIC) model. A, Schematics of the experimental procedure for an eAIC model. B and C, Representative Western blot of LC3-II (microtubule-associated protein light chain 3II) in eAIC and LC3-II quantification (n=4/group). D and E, Tg(GFP-LC3) zebrafish were used to quantify LC3-II induction. Arrows indicate LC3 aggregates (n=3/group). F and G, Autophagy activators exert therapeutic effects, and autophagy inhibitors exert detrimental effects on eAIC, as indicated by changes in both mortality and cardiac function. H, The rank of the top 18 FAA drugs based on a composite score of their therapeutic effects based on survival rate, heart rate, heart function, and phenotypes. Mann-Whitney test in C; Kruskal-Wallis test followed by post hoc Tukey test in D, F, and G; Kruskal-Wallis test followed by Bonferroni post hoc test in G. BafA1 indicates bafilomycin A1; DAPI, dihydrochloride; DOX, doxorubicin; FS, fraction shortening; HR, heart rate; LC3, light chain 3; Rapa, rapamycin; and 3-MA,3-methyladenine. |