- Title
-
Ribosomal Proteins Rpl22 and Rpl22l1 Control Morphogenesis by Regulating Pre-mRNA Splicing
- Authors
- Zhang, Y., O'Leary, M.N., Peri, S., Wang, M., Zha, J., Melov, S., Kappes, D.J., Feng, Q., Rhodes, J., Amieux, P.S., Morris, D.R., Kennedy, B.K., Wiest, D.L.
- Source
- Full text @ Cell Rep.
|
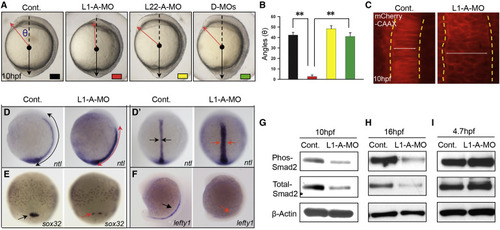
Opposing Roles of RP Paralogs Rpl22 and Like1 in Regulating Gastrulation (A and B) One-cell stage zebrafish embryos were injected with translational-blocking Like1-A-MO (L1-A-MO; 2 ng), L22-A-MO (6 ng), or both (D-MOs), following which effects on gastrulation were assessed. The images of embryos represent lateral views at 10 hpf. The red and black lines indicate the anterior and posterior ends of the body axis, respectively. The angle (θ), which defines the degree of extension, was measured between the red arrow and dashed black line and is represented graphically as the mean ± SD. Control (Cont; black); Like1 MO (L1-A-MO; red); Rpl22 MO (L22-A-MO; yellow); and double-morphants (D-MO; green). Triplicate samples were quantified and the mean ± SD is depicted graphically. ∗∗p < 0.01. (C) Imaging of notochord in Like1 morphants (10 hpf) co-injected with 100 pg mCherry-CAAX mRNA at the one-cell stage. The dorsal view, anterior is at the top. The lateral notochord boundaries are indicated by the dotted yellow lines, and the width of notochord was marked by white lines. (D and D′) Expression patterns of ntl in 10 hpf Like1 morphants. Red arrows mark changes in distribution in the images representing lateral (D) and dorsal (D′) views. (E and F) sox32 and lefty1 expression in Like1 morphants. Red arrows indicate changes in expression or distribution. (E) 75%-epiboly stage, dorsal view. (F) Lateral view, 16 hpf. (G–I) Phospho-Smad2 and total-Smad2 were assessed in 10 hpf (G), 16 hpf (H), and 4.7 hpf (I) Like1 morphants by immunoblotting. All results are representative of at least three experiments performed. See also Figure S1. EXPRESSION / LABELING:
PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|
|
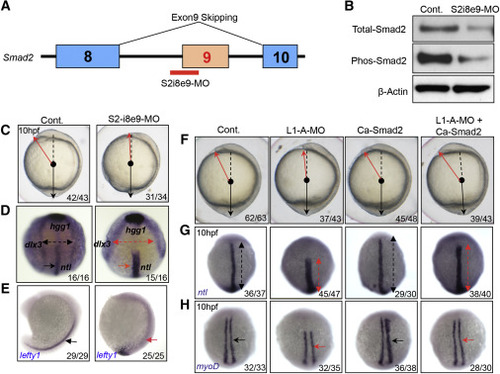
The C&E Defects in Like1 Morphants Can Be Rescued by Re-establishing smad2 Signaling (A) Schematic of the morpholino used to induce exon 9 skipping (S2-i8e9-MO). (B) Immunoblotting of detergent extracts reveals a reduction in total and phospho-Smad2 protein expression in the S2-i8e9-morphants. (C–E) S2-i8e9-MO induction of smad2 mis-splicing phenocopied the C&E defects caused by Like1 knockdown, as indicated by altered morphology (C, red arrow) and alterations in ntl/hgg1/dlx3b and lefty1 expression and distribution, as measured by in situ hybridization (D, 10 hpf, red arrows, anterior dorsal view; E, 16 hpf, lateral view). (F–H) mRNA encoding constitutively activated smad2 (Ca-Smad2, 20 pg) was used for injection alone or co-injected with Like1-A-MO. Embryo morphology (F) as well as the abnormal distribution of ntl (G, red arrows) and myod1 (H, red arrows) at 10 hpf can be rescued by ectopic expression of Ca-Smad2. All embryos are dorsal view with the anterior on top at 10 hpf. All results are representative of at least three experiments performed. See also Figure S3. EXPRESSION / LABELING:
PHENOTYPE:
|
|
The Regulation of smad2 Pre-mRNA Splicing during Gastrulation by Rpl22 and Like1 Is Associated with Their Retention in the Nucleus (A and B) Subcellular location of epitope-tagged Rpl22 and Like1 at 10 hpf (A) and 24 hpf (B). mRNA (100 pg) encoding HA-zRpl22 (HA/L22) and HA-zLike1 (HA/L1) was co-injected with mRNA encoding mCherry-CAAX into one-cell stage embryos and visualized by HA antibody immunostaining. mCherry-CAAX marked the cell membrane, and DAPI marked the nucleus. The red scale bar represents 10 μm. (C) RT-PCR detection of smad2 mis-splicing. Following Like1 MO injection, smad2 mis-splicing was assessed by RT-PCR in 10 and 24 hpf Like1 morphants. All results are representative of at least three experiments performed. See also Figure S4. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|