- Title
-
TAMM41 is required for heart valve differentiation via regulation of PINK-PARK2 dependent mitophagy
- Authors
- Yang, R.M., Tao, J., Zhan, M., Yuan, H., Wang, H.H., Chen, S.J., Zhu, C., de Thé, H., Zhou, J., Guo, Y., Zhu, J.
- Source
- Full text @ Cell Death Differ.
|
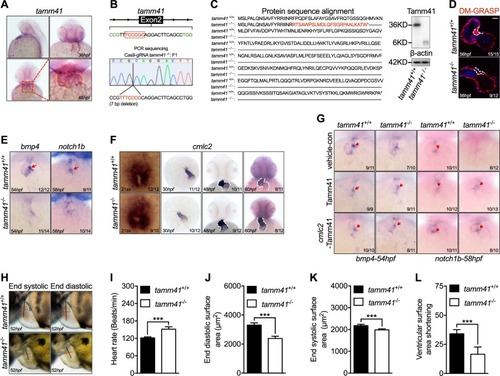
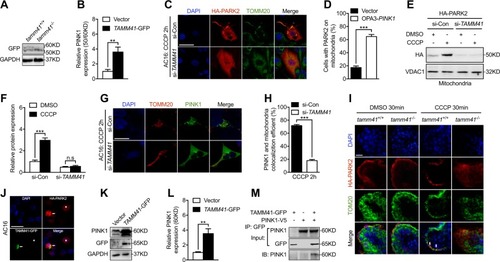
EXPRESSION / LABELING:
PHENOTYPE:
|
|
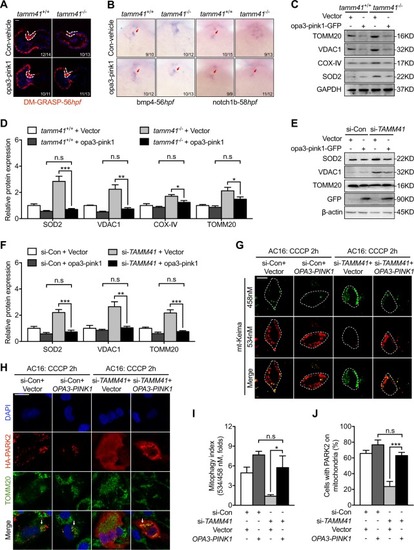
EXPRESSION / LABELING:
PHENOTYPE:
|
|
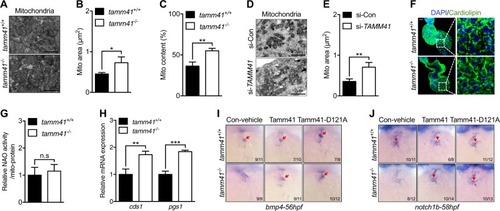
Disturbed mitophagy progression |
|
|
|
|
|
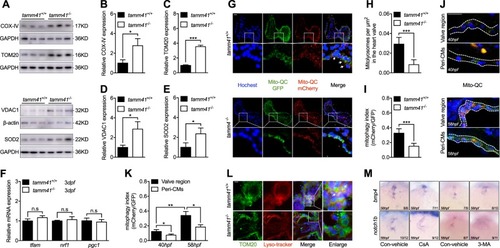
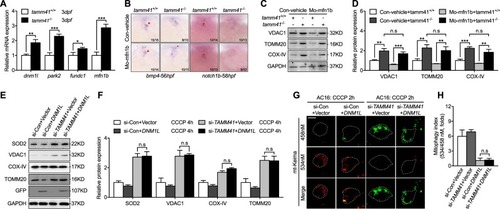
Mitochondria tethered Pink1 overexpression restores |
|
Enforced mitochondrial fission couldn’t restore heart valve malformation with EXPRESSION / LABELING:
PHENOTYPE:
|
|
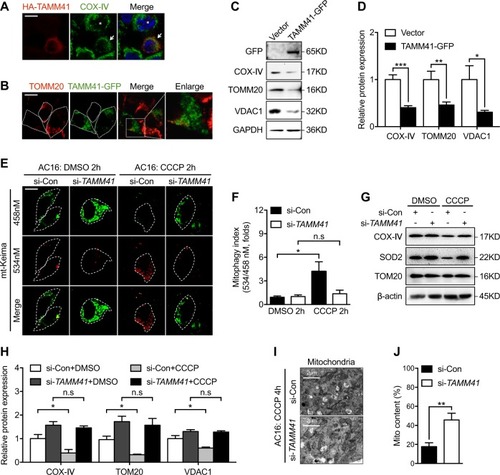
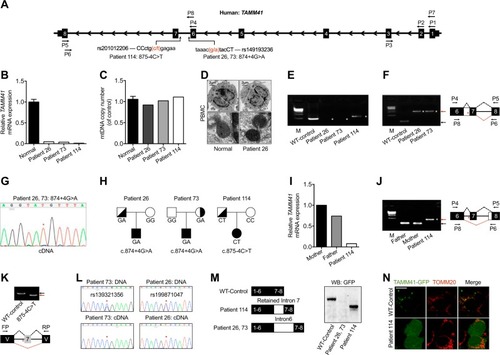
Variations of |