- Title
-
A missense mutation in zbtb17 blocks the earliest steps of T cell differentiation in zebrafish
- Authors
- Lawir, D.F., Iwanami, N., Schorpp, M., Boehm, T.
- Source
- Full text @ Sci. Rep.
|
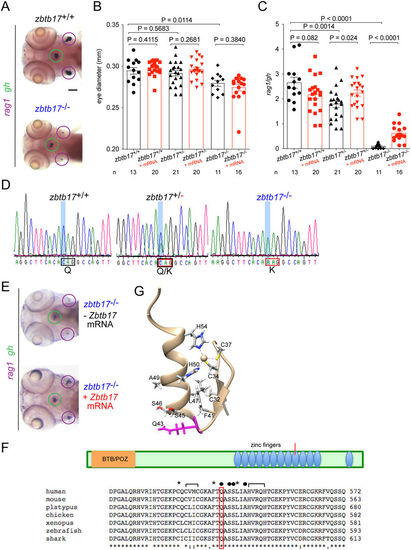
Impaired T cell development in fish homozygous for a missense mutation in zbtb17. (A) Representative microphotographs of wild-type (top panel) and homozygous mutant (bottom panel) zebrafish embryos hybridized with probes specific for rag1 (purple circle) and gh (green circle) at 5 dpf. (B) Eye diameter (means ± s.e.m.) for the three different zbtb17 genotypes (numbers of fish indicated) without (black bars) and with injection of mouse Zbtb17 wild-type mRNA (mRNA) determined at 4 dpf. (C) Thymopoietic indices (means ± s.e.m.) for the three different zbtb17 genotypes without (black bars) and with injection of mouse Zbtb17 wild-type mRNA (mRNA) determined at 4 dpf. (D) Representative sequence traces covering part of exon 10 sequences of wild-type fish (left panel), heterozygotes (middle panel) and homozygous mutants (right panel). The C > A transversion occurs at nucleotide position 1684 of ENSDARG00000074548 (http://www.ensembl.org/Danio_rerio/Transcript/Summary?db=core;g=ENSDARG00000074548;r=23:24525127-24540083;t=ENSDART00000114840). (E) Representative microphotographs of zbtb17 mutant embryos after injection of mouse Zbtb17 mRNA hybridized with probes specific for rag1 and gh at 96 hpf. The thymic rudiments (marked by expression of rag1) are encircled in purple, the hypophysis (marked by expression of gh) is encircled in green. (F) Schematic of the ZBTB17 proteins, indicating the position of known domains, including the 12 zinc finger modules. Below, an alignment of partial protein sequences of vertebrate ZBTB17 proteins around the critical region of the ninth zinc finger (marked in schematic) is shown. The presumptive zinc-coordinating (brackets), DNA-contacting (solid circles) and hydrophobic (asterisks) amino acid residues are marked. Source sequences: human, H. sapiens, Genbank accession number NP_001274532.1; mouse, M. musculus, Genbank accession number NP_033567.2; platypus, Ornithorhynchus anatinus, Genbank accession number XP_003429656.1; chicken, G. gallus, Genbank accession number NP_001263229.1; xenopus, X. tropicalis, Genbank accession number NP_001011141.2; zebrafish, D. rerio, Genbank accession number XP_003201251.1; shark, C. milii, Genbank accession number XP_007905152.1. (G) Structure of the ninth zinc finger of the mouse Zbtb17 protein (source code 2LVT); the mutated Q43 residue is indicated in purple. Scale bar, 100 μm. |
|
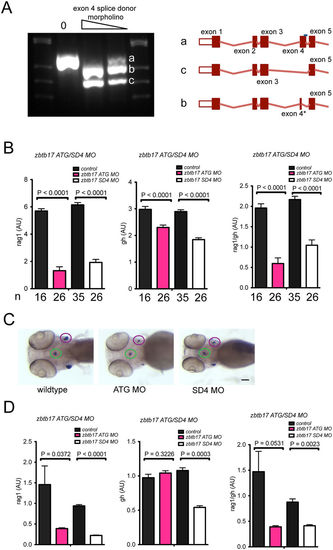
Characterization of zbtb17 morphants. (A) Analysis of aberrant splicing patterns in exon 4 splice donor morphants. Representative pattern indicating wild-type (a) and two aberrant (b,c) cDNAs, whose structures are shown on the right. Isoform b originates from the use of a cryptic splice donor site in exon 4 (nucleotides 763–764 in transcript ENSDART000000114840.3); isoform b represents an exon-skipping event eliminating exon 4. (B) Thympoietic activity measured by RNA in situ hybridization in zbtb17 morphants. The number of embryos in each cohort is indicated. (C) Representative microphotographs of zbtb17 morphants (from (B)) after RNA in situ hybridization with rag1 and gh probes (see Fig. 1). Scale bar, 100 μm. (D) Thymopoietic activity measured by qPCR in zbtb17 morphants at 4 dpf. RNAs were isolated from pools of 20 embryos each. |
|
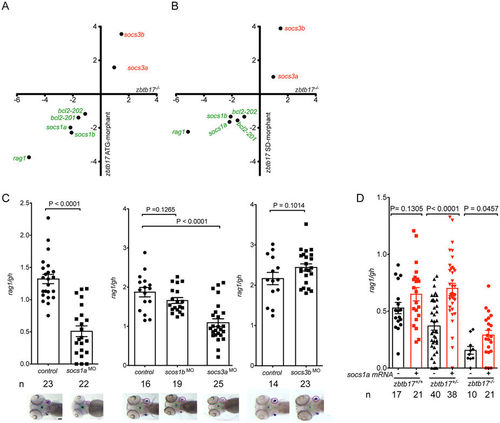
Functional consequences of the zbtb17 missense mutation. (A) qPCR analysis of gene expression levels in homozygous zbtb17 mutants at 5 dpf compared to gene expression levels for zbtb17 ATG-morphants determined at 96 hpf. Expression levels were normalized to those of the actb1 gene and are expressed as fold changes relative to wild-type values; mean values (n = 3 or 4) are shown (detailed results are presented in Supplementary Fig. 1). (B) qPCR analysis of gene expression levels in homozygous zbtb17 mutants at 5 dpf compared to gene expression levels for zbtb17 SD-morphants determined at 96 hpf. Expression levels were normalized to those of the actb1 gene and are expressed as fold changes relative to wild-type values; mean values (n = 3 or 4) are shown (detailed results are presented in Supplementary Fig. 1). (C) Effect of socs gene knock-downs on thymopoietic activity expressed as rag1/gh ratios. Representative RNA in situ hybridization results (the thymic rudiments [marked by expression of rag1] are encircled in purple, the hypophysis [marked by expression of gh] is encircled in green) are shown underneath the relevant bar charts for which mean ± s.e.m. values are shown; each data-point represents one embryos (the total numbers of embryos for each column are indicated). Scale bar, 100 μm. (D) Rescue of the zbtb17 mutant phenotype by provision of socs1a mRNA. The extent of T cell development was assessed by rag1/gh ratios after RNA in situ hybridization without (black bars) and with (red bars) provision of in vitro transcribed socs1a mRNA (the total number of embryos for each column are indicated). |
|
Unperturbed haematopoietic development in zbtb17 mutants. (A) Microphotographs of wild-type and zbtb17 mutant embryos after RNA in situ hybridization with the indicated gene-specific probes and at the indicated times of development (scl, 36 hpf; myb, 36 hpf; gata1, 24 hpf). Scale bar, 100 µm (B) Representative microphotographs of control and morphant embryos on the ikaros:eGFP transgenic background to visualize lymphoid progenitors in the thymus and the caudal haematopoietic tissue (CHT) at 63 hpf. (C,D) Thymus homing assay in control and morphant embryos. Summary statistics of cohorts of embryos (examples in (B)) for cell counts at 63 hpf. |
|
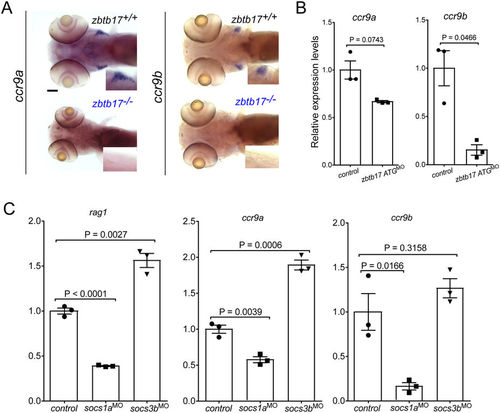
Chemokine receptor expression. (A) Representative microphotographs of wild-type and zbtb17 mutant embryos after RNA in situ hybridization with ccr9a and ccr9b gene-specific probes at 5 dpf. Insets represent 2× magnifications of the thymic regions. No signal is detectable in zbtb17 mutants. Scale bar, 100 μm. (B) Gene expression levels for ccr9a and ccr9b in zbtb17 ATG-morphants as assessed by qPCR. (C) Gene expression levels for rag1, ccr9a and ccr9b in socs1a and socs3b morphants as assessed by qPCR. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |
|
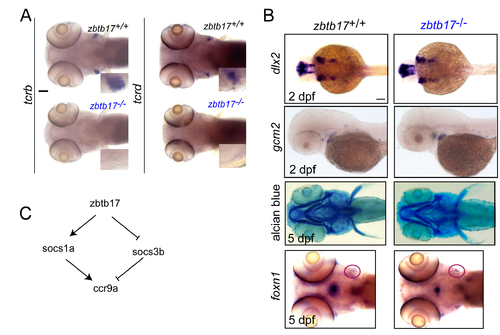
Characterization of zbtb17 mutants. (A) Representative microphotographs of wild-type and zbtb17 mutant embryos after RNA in situ hybridization with tcrb and tcrd gene-specific probes at 5 dpf. Insets represent magnifications of the thymic regions. No signal is detectable in zbtb17 mutants. (B) Microphotographs of wild-type and zbtb17 mutant embryos after RNA in situ hybridization with the indicated gene-specific probes and at the indicated times of development. Alcian blue staining was used to visualize craniofacial cartilaginous structures. (C) Schematic of the network regulating ccr9a expression. Scale bars, 100 μm. |