- Title
-
Sulf1 modulates BMP signaling and is required for somite morphogenesis and development of the horizontal myoseptum
- Authors
- Meyers, J., Planamento, J., Ebrom, P., Krulewitz, N., Wade, E., and Pownall, M.E.
- Source
- Full text @ Dev. Biol.
|
sulf1 is expressed in the developing notochord and trunk musculature in zebrafish embryos. (A–C) Lateral (A) or dorsal (B and C) views of zebrafish embryos fixed at 12 hpf and labeled with antisense probe against sulf1. Expression is strongest in the tailbud and notochord. (D and E) Lateral (D) or ventral (E) views of sulf1 expression in 18 hpf zebrafish embryos. Expression remains strong in the tailbud and notochord, but is also expressed in forebrain, otic vesicle, and somites. (F–H) Expression of sulf1 in 24 hpf zebrafish embryos. (F) Whole mount labeling showing expression in trunk musculature, along with midbrain–hindbrain boundary and optic stalks. (G) Cross-section at approximately the level of the line in (F) showing expression in medial trunk musculature and in the notochord and neural floorplate. (H) Higher magnification view of sulf1 expression in the tail. Scale bars: A, B, D, E, F: 250 μm; C: 25 μm; G: 50 7mu;m; H: 100 μm. EXPRESSION / LABELING:
|
|
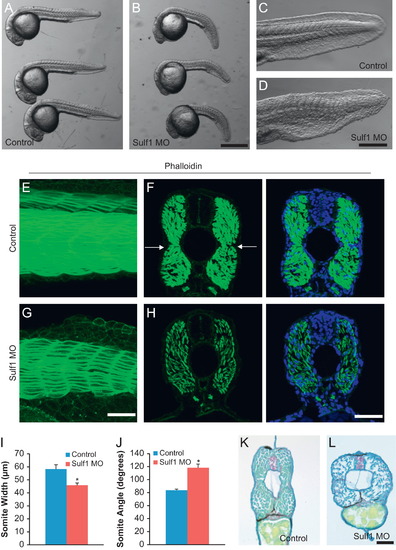
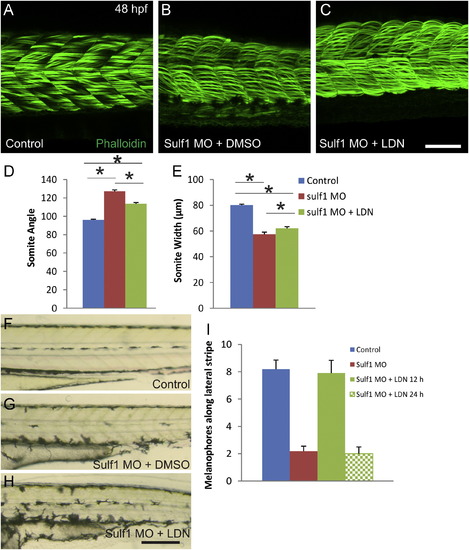
Knockdown of sulf1 leads to somite defects including loss of the horizontal myoseptum. (A) Whole mount view of three fish injected with control morpholino and fixed at 24 hpf, showing normal tail development. (B) Whole mount view of three fish injected with 0.25 pmol sulf1 MO, showing curvature and widening of the tail. (C) Higher magnification view of a control fish at 24 hpf, showing normal chevron-shaped somites. (D) Higher magnification view of a sulf1 morphant fish showing more closely spaced, obtusely angled somites. (E) Lateral view of whole-mount of 48 hpf control zebrafish labeled with 488-conjugated phalloidin. Again normal chevron-shaped somites can be seen. (F) Cross-section of control zebrafish labeled with 488-conjugated phalloidin with arrows marking the distinct horizontal myoseptum that runs along the midline of the fish, counterstained with DAPI. (G) Lateral view of whole-mount of 48 hpf sulf1 morphant zebrafish labeled with 488-conjugated phalloidin, taken at the same exposure and settings as (E). Labeled muscle fibers are much less dense than in controls, and the somites appear rounded, more obtusely angled, and closer together. (H) Cross-section of sulf1 morphant fish at 48 hpf, showing the sparser muscle fibers and the absence of a morphological horizontal myoseptum. (I and J) Quantification of somite width (I) and angle (J) between control and sulf1 morphant fish. Quantification was done on the two somites closest to the proctodeum (approximately somite 15). sulf1 morphants had significantly narrower somites that had a significantly more obtuse angle than control fish (p<0.001 for both, T-test). (K and L) Vibratome cross-section of control (K) or sulf1 morphant (L) zebrafish at 48 hpf stained with hemotoxylin and eosin showing the decreased density of muscle fibers and loss of the horizontal myoseptum in sulf1 morphants. Scale bars: A, B: 500 μm; C, D: 250 μm; E, G: 50 μm; F, H: 50 μm; K, L: 50 μm. |
|
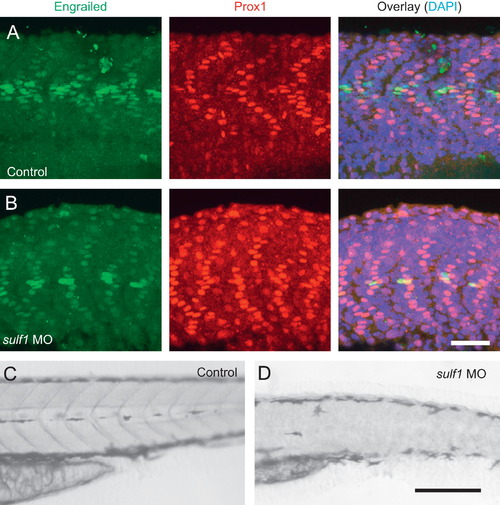
Knockdown of sulf1 leads to decrease in muscle pioneer and medial fast fibers along the horizontal myoseptum. (A and B) Whole mounts of control (A) or sulf1 morphant fish (B) immunolabeled against Engrailed (green) and Prox1 (red), and counterstained with DAPI (blue). Controls have 3–4 muscle pioneers co-expressing Engrailed and Prox1 in each somite, surrounded by a halo of medial fast fibers that express only Engrailed and at weaker levels than the muscle pioneers. In sulf1 morphants, there are only 1–2 muscle pioneers and few medial fast fibers. (C and D) Brightfield view of the mediolateral stripe of melanocytes in control (C) or sulf1 morphant (D) zebrafish. Although control fish have a well-defined stripe of pigmentation that runs along the horizontal myoseptum, these pigment cells are absent in sulf1 morphants. Scale bars: A, B: 50 μm; C, D: 200 μm. |
|
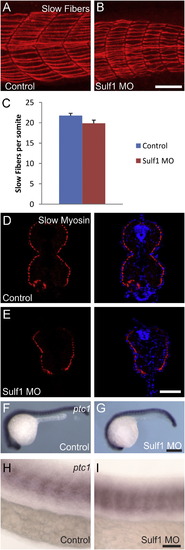
sulf1 morphants have no loss of slow muscle fibers or ptc1 expression. (A and B) Whole-mount labeling of control (A) or sulf1 morphant (B) zebrafish at 24 hpf with the F59 antibody that labels slow muscle fibers at this time-point. Images shown are centered on the 15th somite. Both control and morphant fish have slow muscle fibers along each somite, though the shapes of the somites are different. (C) Quantification of the F59-labeled slow muscle fibers at the 15th somite for control and sulf1 morphant fish revealing that there is a slight, but not statistically significant reduction in the number of slow fibers in sulf1 morphants. (D and E) Cross-sections of control (D) and sulf1 morphant (E) fish at 24 hpf labeled with the BA-D5 antibody that labels slow muscle myosin, showing that the slow muscle fibers in morphants are in approximately the same number and position as controls, though there is not an obvious indentation at the myoseptum in morphants. (F–I) In situ hybridization against the hedgehog target ptc1 in control (F and H) and sulf1 morphant zebrafish (G and I) at 24 hpf. Both control and morphant fish have grossly similar levels and patterns of ptc1 expression. At higher magnification along the yolk extension (H and I), both controls and morphants have equivalent levels of ptc1 in the anterior portion of each somite. The somites are of different shape, but there is no dramatic change in the pattern of ptc1 expression. Scale bars: A, B: 50 μm; D, E: 50 μm; F, G: 250 μm; H, I: 50 μm. |
|
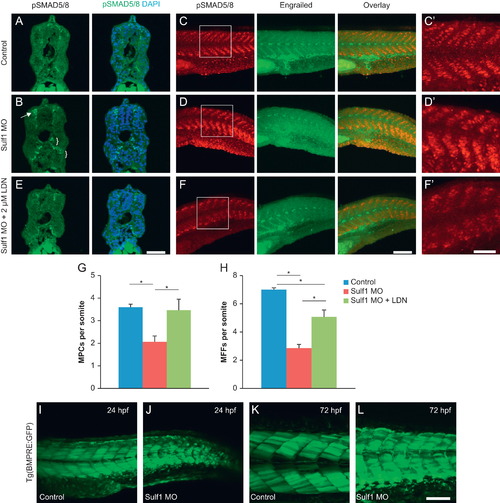
Knockdown of sulf1 in embryonic zebrafish leads to an increase in BMP signaling that can be rescued with inhibition of BMPRI. (A and B) Cross-sections through 36 hpf control (A) or sulf1 morphant (B) zebrafish labeled with anti-pSmad1/5/8 (green) and counterstained with DAPI. sulf1 morphants show stronger immunoreactivity against pSmad1/5/8 particularly around the spinal cord (arrow) and in the ventral regions of the somitic muscle near the notochord (brackets). (C and D) Whole-mount control (C), or sulf1 morphant (D) zebrafish fixed at 24 hpf, and double-labeled for pSmad1/5/8 and engrailed, with images taken at the level around and just posterior to the proctodeum. Control fish show discrete labeling of pSmad reactive nuclei in each somite, but absent from the horizontal myoseptum, where the engrailed-positive muscle pioneers can be found. sulf1 morphants show an increase in the level of pSmad immunoreactivity, and less organization of the pSmad immunoreactive cells particularly in the ventral portion of the more posterior somites, and with less separation between the dorsal and ventral halves of the domains of expression. Again, the number and intensity of engrailed-positive muscle pioneers is reduced in sulf1 morphants. (C′–D′) Magnified view of pSmad immunoreactivity from the boxed regions indicated in C and D. (E and F) Treatment of sulf1 morphant fish with 2 µM LDN193189 beginning at 12 hpf abrogates the increase in pSmad labeling, in cross-sections (E) and whole-mounts (F; magnified in F′). Treatment of sulf1 morphants with 2 μM LDN193189 is sufficient to reduce the levels of pSmad immunoreactivity, particularly in the ventral domain, consistent with inhibition of BMP signaling, and increase the number and intensity of muscle pioneers and medial fast cells. (G and H) Quantification of the number of muscle pioneer cells (G) and medial fast fibers (H) per somite in control (blue), sulf1 morphants treated with DMSO as a vehicle control (green), and sulf1 morphants treated with 2 μM LDN193189 beginning at 12 hpf. LDN treatment restored the number of MPCs to normal and led to a significant increase in the number of MFFs compared to DMSO treated morphants. (I–L) GFP expression under control of repeated BMP response elements in control (I and K) or sulf1 morphant (J and L) at 24 hpf (I and K) or 72 hpf (K and L). While controls have a distinct absence of GFP along the developing horizontal myoseptum, morphants show a greatly reduced gap between the dorsal and ventral expression, with some somites showing strong BMP activity along the lateral midline. Scale bars: A, B, E: 50 μm; C, D, F: 100 μm; C′, D′, F′: 50 μm; I–L: 100 μm. EXPRESSION / LABELING:
PHENOTYPE:
|
|
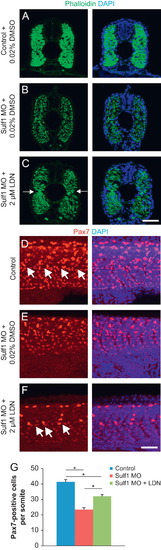
sulf1 morphants have a loss of dermomyotome cells that can be partially rescued by BMP inhibition. (A–C) Cross-sections of control fish (A), sulf1 morphants treated with DMSO as a vehicle control (B) or sulf1 morphants treated with 2 μM LDN193189 (C) beginning at 12 hpf. Fish was fixed at 48 hpf and labeled with phalloidin to label actin-rich muscle fibrils. While sulf1 morphants have a decrease in the density of muscle fibers and lack a defined horizontal myoseptum compared to controls, the density of muscle fibers and myoseptum are partially rescued in morphants treated with LDN to decrease BMP signaling. (D–F) Whole-mount views of control (D), sulf1 morphant (E), and sulf1 morphant fish treated with 2 μM LDN193189 (F) fixed at 24 hpf and labeled with an antibody against Pax7, which strongly labels neural crest derived cells and also labels dermomyotome cells. Images are taken around the proctodeum (approximately somite 15). (D) Control fish have Pax7-expressing dermomyotome cells throughout each somite, with a distinct population of elongate cells along the developing horizontal myoseptum (arrows). (E) sulf1 morphants have fewer Pax7-expressing dermomyotome cells across the somite, and in particular have no elongate cells running along the horizontal myoseptum. (F) Treatment of sulf1 morphants with LDN193189 increases the number of Pax7-expressing dermomyotome cells in each somite, including restoring the population of elongate cells along the horizontal myoseptum (arrows). (F) Quantification of the number of Pax7-expressing dermomyotome cells in each somite (excluding strongly labeled neural crest cells), from control (blue), sulf1 morphants treated with DMSO (red) and sulf1 morphants treated with 2 μM LDN193189 beginning at 12 hpf (green). Statistical significance is shown by asterisks (p<0.001 for each comparison; ANOVA). Scale bars: A–C: 50 μm; D–F: 50 μm. |
|
Inhibition of BMP signaling in sulf1 morphants partially rescues somite shape and also restores the lateral stripe of pigmentation, but only if done during somitogenesis. (A–C) Phalloidin labeled whole-mounts of 48 hpf control fish (A), sulf1 morphants treated with DMSO as a vehicle control (B), or sulf1 morphant zebrafish treated with 2 μM LDN193189 beginning at 12 hpf (C). While the sulf1 morphants have somites that are more narrow, rounded and obtuse compared to controls, with smaller and less organized actin fibers, inhibition of BMP signaling with LDN193189 partially rescues the phenotype. The LDN-treated somites appear to be less rounded, and have a reduction in the angle compared to untreated or DMSO-treated morphants. (D and E) Quantification of somite angle (D) and width (E) in control (blue), sulf1 morphants treated with DMSO (red) and sulf1 morphants treated with 2 μM LDN193189 (green) beginning at 12 hpf. While sulf1 morphants have significantly more obtuse and more narrow somites than controls (p<0.001; ANOVA), treatment with LDN leads to a significant decrease in the somite angle (p<0.001; ANOVA) and slight, but significant increase in the width of each somite (p<0.05; ANOVA). (F–H) Brightfield views of 72 hpf control (F) or sulf1 morphant zebrafish treated with DMSO (G) or 2 μM LDN193189 (H) at 12 hpf, showing location and number of melanocytes along the lateral stripe. (I) Quantification of the number of melanocytes along the lateral stripe of control (blue), sulf1 morphants treated with DMSO at 12 hpf (red), sulf1 morphants treated with 2 μM LDN193189 at 12 hpf (solid green), and sulf1 morphants treated with 2 μM LDN at 24 hpf (green checked). sulf1 morphants have a significant decrease in the number of melanocytes along the lateral stripe (p<0.001 compared to control; ANOVA), which can be fully rescued by BMP inhibition with LDN treatment beginning at 12 hpf (p<0.001 compared to DMSO-treated; p>0.05 compared to control; ANOVA). Treatment with LDN at 24 hpf does not rescue the number of melanocytes (p>0.05 compared to DMSO-treated; ANOVA). Scale bars: A–C: 100 μm; F–H: 200 μm. PHENOTYPE:
|
|
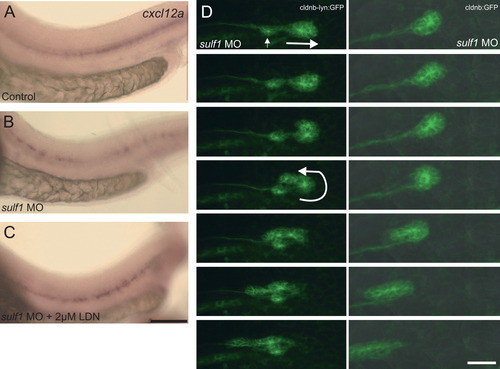
sulf1 knockdown leads to improper migration of the lateral line primordium and a change in cxcl12a (sdf1a) expression along the lateral midline, which can be rescued by inhibition of BMP. (A–C) In situ hybridization for cxcl12a (sdf1a) in control fish (A), sulf1 morphant fish (B), or sulf1 morphant fish treated at 12 hpf with 2 μM LDN19189 (C). In the control fish, the cxcl12a expression is in a stripe along the lateral midline. In sulf1 morphants, the cxcl12a expression is greatly reduced with gaps in the expression along the lateral midline. Treatment with 2 μM LDN beginning 12 hpf can rescue the intensity and positioning of cxcl12a expression. (D) Individual frames from two separate time lapse series of sulf1 morphant Tg(cldnb:lynGFP) zebrafish starting at <32 hpf with subsequent frames stacked vertically being 30 min apart. In both examples, the small primordium has recently deposited a neuromast and is moving toward the tail (right arrow). By the third frame the primordium begins to turn and completely turns itself around to reverse direction (curved arrow), and it then proceeds back toward the head. Scale bars: A–C: 100 μm; D: 50 μm. |
|
sulf1 knockdown leads to loss of FGF signaling within the lateral line primordium but can be partially rescued by BCI. (A) Whole mount Tg(cldnb:lyngfp) zebrafish at 54 hpf showing normal distribution of posterior lateral line (pLL) neuromasts. (B) Whole mount Tg(cldnb:lynGFP) sulf1 morphant zebrafish at 54 hpf showing a decreased number of pLL neuromasts along the tail, quantified in the adjacent graph. (C) Quantification of neuromast number from control and sulf1 morphant zebrafish. Morphants have 4.6±0.6 neuromasts while controls have 7.5±0.3 neuromasts along their trunk. (D) View of the pLL primordium from a control Tg(cldnb:lynGFP) zebrafish at ∼32 hpf shortly after deposition of a neuromast (arrow), where the rosette patterning of the next two neuromasts to be deposited can be seen (arrows). (E) View of the pLL primordium of a Tg(cldnb:lynGFP) sulf1 morphant zebrafish at ∼32 hpf shortly after deposition of a neuromast (arrow). The primordium is much smaller than in controls, and the typical rosette patterns within the neuromast or in the trailing edge of the primordium are not visible. (F and G) In situ hybridization of an FGF10a probe in control (F) or sulf1 morphant (G) zebrafish at 36 hpf. FGF10a is expressed in the leading zone of the primordium, and in the central region of a recently deposited neuromast (arrow) and at the center of the rosette of the next neuromast to be deposited (arrowhead). In sulf1 morphants, FGF10a is expressed more uniformly across the smaller neuromast and is not focally expressed at the center of a rosette along the trailing edge. (H and I) Tg(cldnb:lynGFP) zebrafish treated at 24 hpf with either DMSO (H) or 1 μM FGFRTKi (I) imaged at 54 hpf showing that partial inhibition of FGFR1 can phenocopy the reduced number of deposited neuromasts. (J) sulf1 morphant Tg(cldnb:lynGFP) zebrafish treated at 32 hpf with 2.5 μM BCI and imaged at 54 hpf, showing a decrease in the spacing between the last 4 deposited neuromasts compared with the initial two. (K) Quantification of the distance between the first two deposited neuromasts (anterior) and last two deposited neuromasts excluding the terminal cluster (posterior) at 54 hpf from control fish (blue), sulf1 morphant fish treated with DMSO (red), and sulf1 morphant fish treated at 32 hpf with 2.5 μM BCI (green). BCI had no effect on neuromast distance in the anterior neuromasts that had already been deposited before it was added (p>0.05 compared to DMSO-treated morphants; ANOVA), but restored the appropriate distance between posterior neuromasts deposited after its application (p<0.001 compared to DMSO-treated morphants; p>0.05 compared to control; ANOVA). Scale bars: A, B, H, I, J: 500 μm; D, E: 50 μm; F, G: 50 μm. |
|
|
Reprinted from Developmental Biology, 378(2), Meyers, J., Planamento, J., Ebrom, P., Krulewitz, N., Wade, E., and Pownall, M.E., Sulf1 modulates BMP signaling and is required for somite morphogenesis and development of the horizontal myoseptum, 107-21, Copyright (2013) with permission from Elsevier. Full text @ Dev. Biol.