- Title
-
Genetic defects of pronephric cilia in zebrafish
- Authors
- Zhao, C., and Malicki, J.
- Source
- Full text @ Mech. Dev.
|
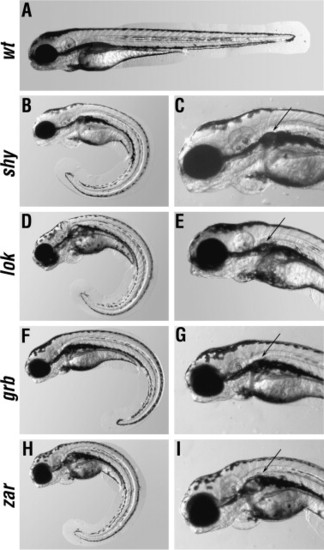
External phenotypes of mutant animals. Lateral views of wild-type (A) and mutant (B–I) larvae at 3 dpf. (C, E, G, and I), show high magnifications of the anterior region of mutant larvae. All mutants shown develop kidney cysts (arrows in C, E, G, and I) and ventrally curled tails (B, D, F, and H). There are no obvious differences in eye and ear phenotypes between mutant and wild type larvae. In all panels, anterior is left, dorsal is up. PHENOTYPE:
|
|
Histological analysis of the anterior pronephros. Transverse sections through the glomerulus (arrow) and the pronephric tubule (asterisks) in mutant and wild type embryos at 5 dpf. The triangle in (A) indicates the pronephric duct. All mutant strains display a distended pronephric tubule lumen (asterisks in B–E). The glomerulus is stretched out dorso-ventrally in the mutants (arrow in B–E). Enlargements of glomeruli (B′–E′) correspond to mutant strains shown in (B–E). In all panels, dorsal is up. PHENOTYPE:
|
|
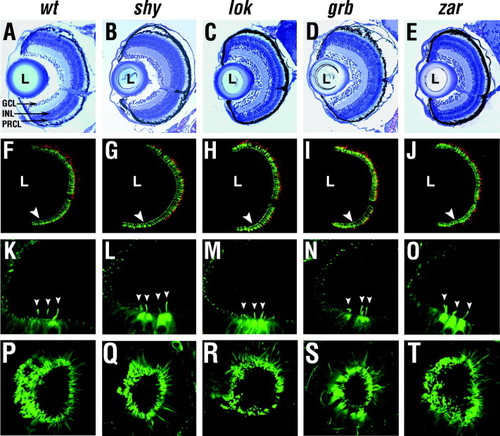
Phenotype of sensory neurons. (A–E) Transverse plastic sections through the central retina near the optic nerve at 7 dpf. (F–J) Confocal images of transverse frozen sections through the retina near the optic nerve stained with Zpr1 (green) and anti-blue opsin (red) antibodies at 7 dpf. (K–O) Anterior tether cells (arrowheads) in otic vesicles immunostained for acetylated-α-tubulin at 30 hpf. (P–T) Nose cilia stained for acetylated-α-tubulin at 3 dpf. In A–J, “L” indicates the lens, midline is right, dorsal is up. Arrowheads in (F–J) indicate the photoreceptor cell layer. GCL, ganglion cell layer; INL, inner nuclear layer; PRCL, photoreceptor cell layer. PHENOTYPE:
|
|
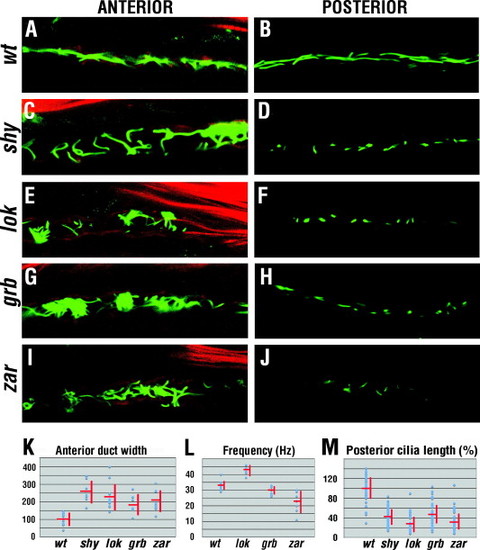
Phenotypes of pronephric cilia. (A, C, E, G, and I) Anterior kidney duct visualized with anti-acetylated tubulin antibody (green) and phalloidin (red) at 30 hpf. Compared to those in the wild type, mutant cilia in this region are misdirected. (B, D, F, H, and J) Posterior pronephric duct cilia visualized by anti-acetylated tubulin antibody (green) at 30 hpf. In mutants, cilia are misdirected and their length is decreased. (K) Dot graph of kidney duct width measurements. (L) Frequency of anterior kidney duct cilia movement. (M) Cilia length in the posterior kidney duct of mutant strains, relative to the wild-type, which is set at 100%. In (A–J), dorsal is up. |
|
Left–right asymmetry phenotype in grbtm304 mutant embryos. (A–C) Ventral views of wild-type and grbtm304 embryos at 72 hpf. Wild-type animals display normal heart looping phenotype (A). Heart looping is frequently absent (B) or inverted (C) in grbtm304 mutant embryos. (D–F) In situ hybridization staining for a heart marker, cmlc2, at 3 dpf. Normal heart orientation in the wild type is shown in (D). The absence of obvious heart looping (E) or reverse looping (F) in mutant embryos. (G–I) The expression of lefty1 in embryos collected from crosses between grbtm304/+ heterozygotes. The lefty1 transcript is expressed on the left side of neural tube in the majority of embryos (G). The lefty1 expression domain extends to both sides (H) or is absent (I) in some embryos. (J, K, and L) The expression of lefty2 in embryos collected from crosses between grbtm304/+ heterozygotes. (J) The normal expression of lefty2 transcript on the left side of the lateral plate mesoderm. The lefty2 expression domain shifts to the right side (K) or is absent (L) in some embryos. A, atrium; V, ventricle. In all panels anterior is up. Arrowhead indicates the midline. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Phenotype of Kupffer’s vesicle. Kupffer’s vesicle (KV) cilia visualized with anti-acetylated tubulin antibody at the 10-somite stage. (A) KV cilia of a typical embryo collected from a control cross between wild-type animals. (B–D) KV cilia of embryos from crosses between two mutant heterozygotes of shy (B), grb (C), and zar (D). Individuals with roughly the average number of cilia are shown. (E, F, H, and I) Phenotypes of KV cilia in two typical embryos collected from a cross between lokto237b TU/WIK heterozygotes (E,F) or lokto237b TU/AB heterozygotes (H,I). In comparison to presumptive wild-type individuals from the same genetic background (E,H), lokto237b mutant embryos display shorter KV cilia (F,I). (G) The number of KV cilia in wild-type and mutant embryos. (J) A graph of KV cilia length in mutant lines relative to the wild type, which is set to 100%. Each dot represents the average length of at least 9 cilia in one embryo. Horizontal and vertical bars in (G,J) indicate the average and the standard deviation respectively. As length measurements in (J) appear to form two clusters, which presumably represent homozygous mutant and phenotypically wild-type embryos, the average and the standard deviation are provided for each cluster separately. TW, Tuebingen/WIK genetic background; TA, Tuebingen/AB genetic background. Embryos in panels (A–D) were of homozygous Tuebingen background. PHENOTYPE:
|
|
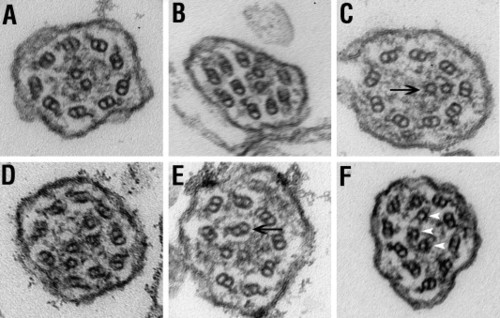
Ultrastructure of cilia. Images of ultrathin cross-sections through the pronephric cilia of wild-type (A) and lokto237b (B–F) mutant embryos at 5 dpf. (A) Typical 9 + 2 microtubule configuration in wild-type animals. (B–F) Abnormal arrangements of microtubules in lokto237b mutant embryos frequently feature misplaced microtubules (arrows in C and E) or supernumerary microtubule singles (arrowheads in F). Note the irregular shape of membrane in some mutant cilia (B,F). PHENOTYPE:
|
Reprinted from Mechanisms of Development, 124(7-8), Zhao, C., and Malicki, J., Genetic defects of pronephric cilia in zebrafish, 605-616, Copyright (2007) with permission from Elsevier. Full text @ Mech. Dev.