- Title
-
Harmonin (Ush1c) is required in zebrafish Muller glial cells for photoreceptor synaptic development and function
- Authors
- Phillips, J.B., Blanco-Sanchez, B., Lentz, J.J., Tallafuss, A., Khanobdee, K., Sampath, S., Jacobs, Z.G., Han, P.F., Mishra, M., Williams, D.S., Keats, B.J., Washbourne, P., and Westerfield, M.
- Source
- Full text @ Dis. Model. Mech.
|
Zebrafish ush1c is expressed in retina and the inner ear. (A) The predominant splice form of ush1c corresponds to mammalian isoform A, with 21 exons that produce a 1.6 kb mRNA. A hydrophobicity plot of zebrafish (red) and human (blue) isoform A protein sequence shows conserved residue polarity. % amino acid identity with human protein is indicated below the plot. The positions of the ush1cfh293 nonsense mutation (red arrowhead), the MO target (green arrowhead) and the region recognized by the zebrafish harmonin antibody (red bar) are indicated. (B) At 5 dpf, ush1c transcripts are abundant in the inner retina (black arrow), the sensory patches of the ear (white arrows), the intestinal tract (i) and the swim bladder (sb). Fainter expression in the brain (b) and trunk muscles (m) is also observed. (C) High-magnification view of a 5 dpf whole-mount zebrafish ear showing ush1c expression in all sensory patches (ac, lc, pc: anterior, lateral and posterior cristae, respectively; am: anterior macula) and an adjacent head neuromast (n). (D) ush1c transcripts are restricted to cells in the inner nuclear layer in 5 dpf sectioned retina. (E) Central section of an adult zebrafish retina showing continued strong expression in the inner retina; expression is detected in the outer nuclear layer as well. onl, outer nuclear layer; inl, inner nuclear layer; gcl, ganglion cell layer. Scale bars: 100 μm (B); 20 μm (C); 50 μm (D,E). EXPRESSION / LABELING:
|
|
Mutation and morpholino knockdown of ush1c produce behavioral defects. (A) RT-PCR of control and splice-blocking morpholino-injected embryos from 1 to 5 dpf reveals a smaller splice form lacking exon 2 that predominates for the first 4 days of development and persists through the fifth day. (B) Western blots from extracts of control + ush1c MO-injected embryos from 3 to 5 dpf (left) and ush1cfh293 homozygous mutant embryos alongside wild-type siblings (right) confirm morpholino efficacy and antibody specificity. Samples from 3 dpf to 5 dpf control larvae probed with the zebrafish harmonin antibody show a 60 kDa band consistent with the predicted size of isoform A. Additional smaller bands are visible faintly in the 4 dpf control and are more pronounced and abundant in the 5 dpf control. The band corresponding to the isoform A variant is undetectable in 3 dpf and 4 dpf morphant samples. The major band reappears at 5 dpf at a slightly smaller weight, and there is a reduced number of smaller bands in this sample. In extracts from 5 dpf ush1cfh293 homozygous mutants, no bands are observed. Extracts from wild-type siblings from the same clutch show the characteristic banding pattern of the 5 dpf uninjected controls, allowing for the slightly longer running time for this gel. A pan-actin antibody was used as a loading control. (C) Behavioral assays of control, mutant and morpholino-injected larvae show (top) impaired startle response and balance, and (bottom) reduced optokinetic response, tracking an average of 50% or fewer of the dark stripes (average targets tracked for wild type: 11.5±1.27; ush1c mutants: 6.13±2.78, P<0.0001; ush1c MO: 5.13±2.85, P<0.0001; n=30 for each group). PHENOTYPE:
|
|
Harmonin is required in otic sensory patches for normal bundle development. (A–C) Phalloidin-stained F-actin (green) and anti-zebrafish harmonin antibody labeling (red) in 80-hpf wild-type ears show localization of harmonin relative to the stereocilia in the five otic sensory patches (ac, anterior crista; am, anterior macula; lc, lateral crista; pc, posterior crista; pm, posterior macula) and in the neuromasts (n). The anti-harmonin antibody signal is reduced in sensory patches and in neuromasts in ush1c mutants (B) and ush1c-MO-injected larvae (C). (D–L) High-magnification view of phalloidin-stained hair bundles in the anterior and posterior maculae of 5 dpf larvae. Hair bundle number is reduced and organization of the existing bundles is disrupted in mutant (G–I) and morphant (J–L) anterior (G,J) and posterior (H,K) maculae, compared with the anterior and posterior maculae of wild-type controls (D and E, respectively). Closeup images of hair bundles of the anterior maculae (F,I,L) show bent and splayed bundles in mutants and morphants. For mutant analysis, anterior macular wild-type average=54.6±3.5 bundles; n=8; ush1c mutant average=33±3.5; n=7; P<0.0001. Posterior macula wild-type average=68.2±2.6 bundles; n=9; ush1c mutant average=40.5±4.2; n=10; P<0.0001. For the morpholino experiment, anterior macula control average=41.8±4.8 bundles; MO average=21.8±3.8; n=5 for each group; P<0.0001. Posterior macula control average=71.8±4.6 bundles; MO average=48.2±6.9; n=5 for each; P<0.0001, 5 dpf. Scale bars: 50 μm (A–C); 10 μm (D–L). EXPRESSION / LABELING:
PHENOTYPE:
|
|
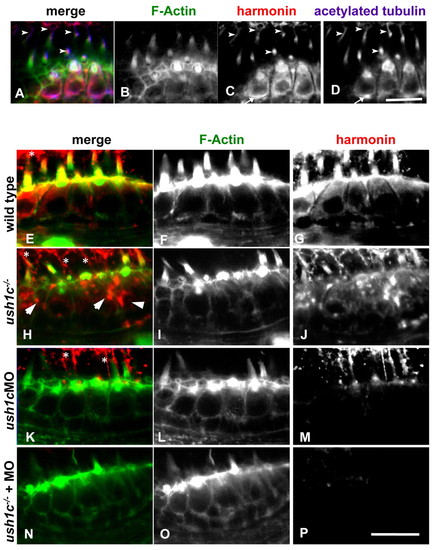
Subcellular localization of harmonin in mechanosensory hair cells. Co-labeling of wild-type 5 dpf anterior macula hair cells (A–D) shows anti-zebrafish harmonin localization (red) in the hair bundles (green), kinocilia (arrowheads), cell bodies and at synaptic boutons (arrow in C and D). Incubation conditions favoring hair bundle visualization (E–P) show strong colocalization of harmonin within the bundle structure as well as in the cuticular plate in 5 dpf wild types (E–G). Kinocilia labeling can be seen above the bundle region (asterisks). Enhanced signal detection in mutant hair cells (H–J) shows localization persisting in the kinocilia and in existing bundle structures. Kinocilia are more visible in these panels owing to the reduced bundle architecture in the mutant. Protein distribution seems globular and disorganized in the cell bodies (arrowheads). Hair cells of ush1c MO larvae (K–M) show strong knockdown of harmonin, with localization persisting only in kinocilia at this stage, and no signal observed within hair bundles. Injecting the morpholino into the ush1c-/- mutants reduces subcellular localization of harmonin to undetectable levels in hair cells (N–P). Scale bars: 10 μm. |
|
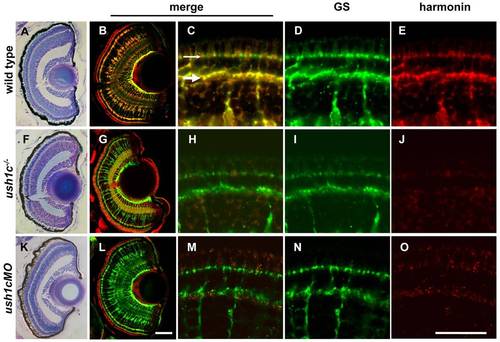
Harmonin localization in the larval retina is restricted to Müller glia. Semi-thin plastic sections stained with toluidine blue show consistent size, lamination and organization of 5 dpf wild-type, mutant and morphant retinas (A,F,K). 16-μm larval retina sections (B,G,L) show colocalization of the Müller cell marker glutamine synthetase (GS; green) and anti-zebrafish harmonin (red) in wild type (B). Both antibodies label Müller cell processes throughout the retina. Anti-zebrafish harmonin labeling is reduced in mutant (G) and morphant (L) Müller cells. Müller cell number and morphology is unaffected. Nonspecific signal associated with photoreceptor outer segments is observed in all panels at this magnification. See supplementary material Fig. S4 for comparison. High-magnification images of anti-zebrafish harmonin and GS labeling in Müller cell processes at the OPL (large arrow) and OLM (small arrow) of the 5 dpf retina are shown (wild type, C–E; mutant, H–J; morphant, M–O). Scale bars: 50 μm (A,B,F,G,K,L); 20 μm (C–E,H–J,M–O). |
|
Harmonin localizes in Müller cells through all zebrafish life stages and is evolutionarily conserved. (A–F) Localization of anti-zebrafish harmonin (red) in Müller cells persists in adult zebrafish retinas. Some low level signal is sometimes observed in photoreceptor synapses (arrow in A) and in photoreceptor outer segments (arrow in D). The strongest signal within the photoreceptor layer is associated with Müller cell processes projecting into the subapical region (asterisks). Nonspecific fluorescence of photoreceptor outer segments at this stage can be compared with panel H in supplementary material Fig. S4. (G–L) Anti-mouse harmonin (SDI; G,I) and anti-human harmonin (Santa Cruz; J,L) detect localization in photoreceptors and Müller cells [glutamine synthetase (GS) is labeled in green; harmonin antibodies in red] in retinas from P10–P11 mouse pups. The outer nuclear layer (onl), inner nuclear layer (inl) and ganglion cell layer (gcl) are labeled in merged panels. Scale bars: 50 μm (A–C,G–L); 20 μm (D–F). EXPRESSION / LABELING:
|
|
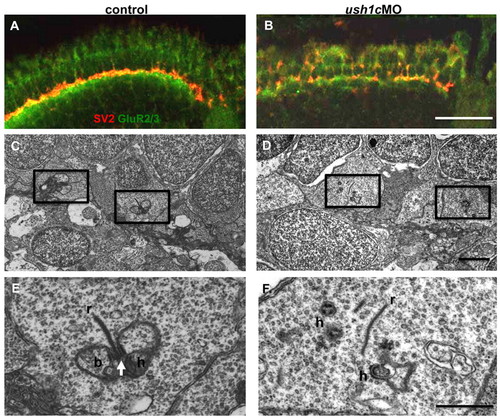
Synaptic maturation is impaired in ush1c morphants. (A,B) Anti-SV2 (red) and -GluR2/3 (green) antibodies mark the pre-and postsynaptic regions of the OPL, respectively. Tightly associated localization of these factors is apparent in the control retina (A), and is notably disrupted in the morphant retina (B) at 5 dpf. (C–F) Electron micrographs of cone pedicles in 6 dpf control and morphant larvae. In control retinas (C,E), the presence of multiple triads (boxed areas), consisting of a synaptic ribbon (r), arciform density (arrow) and postsynaptic processes from bipolar (b) and horizontal (h) cells, indicate normal synaptic maturation. An enlargement of a triad is shown in E. In morphant retinas (D,F), fewer triads are present (boxed areas in D; enlarged in panel F), and floating ribbons (r) and distant postsynaptic processes (h) are observed in cone pedicles. Scale bars: 20 μm (A,B); 1 μm (C,D); 500 nm (E,F). |
|
ush1c mRNA is reduced in ush1cfh293 homozygous mutant tissues. (A-D) 5′ ush1c riboprobe expression in the ear (A,B) and gut (C,D) of 5 dpf whole larvae. Specific ear expression is undetectable in mutants, but some residual transcript remains in the gut tissue. (E-F) Anti-zebrafish harmonin antibody labeling (red) is abundant in sectioned 5 dpf gut tissue of a wild-type sibling, but undetectable in an ush1c mutant on the same slide. Gut autofluorescence is shown in green to indicate gut tissue morphology. Scale bars: 50 μm, A-D; 100 μm, E,F. |
|
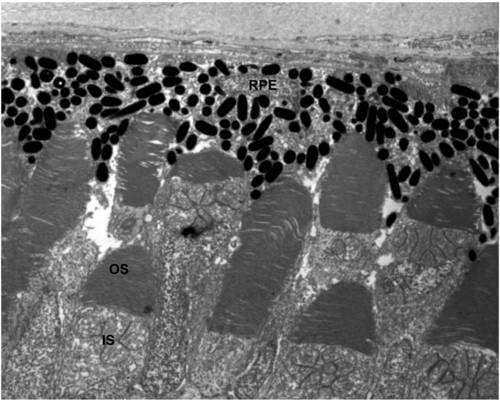
Organization of outer segment disk membranes appears normal in 6dpf ush1cMO larvae. A Transmission electron micrograph showing photoreceptor outer segment ultrastructure in morphant larvae. RPE: Retinal Pigmented Epithelium; OS: Outer Segment; IS: Inner segment. Scale bar: 1μm. |
|
Anti-zebrafish harmonin localization in 6 and 8dpf retinas. (A) Bright, even labeling is seen in Müller glial cells throughout the retina in 6d wild-type animals. (B) Müller cells in 6d morphant animals are faintly labeled in the central retina, but more brightly labeled adjacent to the ciliary marginal zones (arrows). (C) No Müller cell labeling is observed in stagematched ush1c homozygous mutant retinas. (D) A stage matched wild-type embryo treated with blocking solution provides a control for autofluorescence of the photoreceptor outer segments and plexiform layers. (E) Harmonin in 8dpf retinas shows persistent staining in Müller cells and no detectable photoreceptor staining above background levels. Harmonin localization in Muller cells of 8dpf mutants is diminished (F) to background levels. Panel G shows an 8d retina processed with the ABC and TSA kits without incubation in primary antibody to demonstrate the nonspecific signal from synaptic and photoreceptor outer segment membranes. Panel H shows autofluorescence in a 6 mo adult retina incubated with blocking solution only. All negative control images were captured with the same exposure settings as the experimental panels to which they are compared. (I): graphs of retinal cell death by cell layer in 5 and 8dpf larvae. Asterisks denote statistical significance. Scale bars: A-D, H 50 μm; E-G, 10 μm |

Unillustrated author statements PHENOTYPE:
|