- Title
-
Three modules of zebrafish Mind bomb work cooperatively to promote Delta ubiquitination and endocytosis
- Authors
- Chen, W., and Casey Corliss, D.
- Source
- Full text @ Dev. Biol.
|
Neuronal progenitors in Hi904 mutants differentiate prematurely to cause supernumerary primary neurons at the expense of glia and secondary neurons. (A) BrdU incorporation (gray) and expression of pan-neuronal marker HuC (brown) in cervical spinal cord show premature cell cycle exit and differentiation in the mutant. (B) Anti-Islet1/2 staining of Rohon-Beard neurons (arrow) and primary motoneurons (arrowhead) in trunk spinal cord indicates supernumerary and disorganized primary neurons in the mutant. (C) Zn5 antibody staining of secondary motoneurons (arrowhead) and lateral longitudinal fasciculus (arrow) in trunk spinal cord show the absence of secondary neurons in the mutant. (D) Zrf1 staining of radial glial cells in trunk spinal cord reveals the absence of glial cells in the mutant. |
|
Hi904 mutation diminishes Notch signaling. (A–E) In situ hybridization with antisense riboprobes of Notch pathway components in embryos at 14 hpf indicates increase of deltaA expression (A), notch1a expression is not significantly different (B), diminished her4 expression (C), and enhanced neurogenin-1 (D) and islet-1 expression (E) in the mutants. (F) Expression of HuC after injection of synthetic RNA encoding the Myc-tagged Notch1aICD on one side (marked gray with anti-Myc immunostain) shows blockage of neurogenesis on the injected side in both wild-type and mutant embryos. The inserts are blowups of a part of the images. |
|
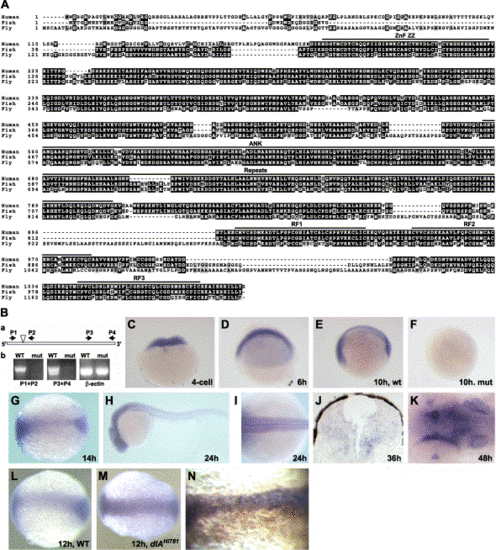
Hi904 mutation disrupts a conserved putative E3 ubiquitin ligase that is highly expressed in the CNS. (A) Alignment of amino acid sequence for human, zebrafish, and fly KIAA1323 orthologs shows highly conserved regions. (B) A schematic diagram of Hi904 mRNA indicates the location of the viral insert, as well as the location and orientation of PCR primers used for RT-PCR analysis (a). RT-PCR analysis of wild-type and mutant RNA at 36 hpf shows the absence of Hi904 mRNA, but not β-actin RNA in mutants (b). (C–K) Expression of Hi904 mRNA measured by in situ hybridization in wild-type embryos at four-cell stage (C), 6 hpf (D), 10 hpf (E), 14 hpf (G), 24 hpf (H and I), 36 hpf (J), and 48 hpf (K) and in Hi904 mutant embryo at 10 hpf (F). Expression at 48 hpf (K) is prominent in the inner ears (arrow), tectum (white arrowhead), and optic stalk (black arrowhead). (L, M) Expression of Hi904 mRNA in wild-type embryos (L) and dlAhi781 mutants (M) at 12 hpf shows an increase of Hi904 in deltaAhi781 mutants. (N) Expression of Hi904 mRNA in wild-type embryos after injection of synthetic RNA encoding zebrafish DeltaA (marked brown by immunostaining of co-injected β-gal) that suppresses Hi904 expression. Thin line indicates midline. ANK, ankyrin domain; RF, ringer finger domain; ZZ ZnF, ZZ zinc finger domain, retroviral insertion; P, primer; β-act, β-actin. |
|
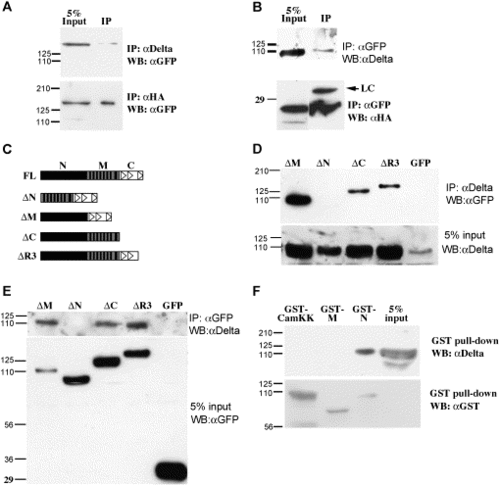
Mib is associated with Delta in Cos7 cells. (A, B) Western blot analysis of co-immunoprecipitation samples reveals the association between full-length Mib with DeltaD (upper panel) and HA-DeltaAICD (lower panel). (C) Schematic diagram of the Mib portion of EGFP fusion proteins. The solid vertical bars indicate the ankyrin repeats, the empty triangles indicate the ring fingers. (D, E) Co-IP analysis of Mib deletions and DeltaD shows that the N-terminal half is necessary for DeltaD association. (F) GST pull-down analysis shows that the N-terminal half is sufficient to associate with DeltaD. |
|
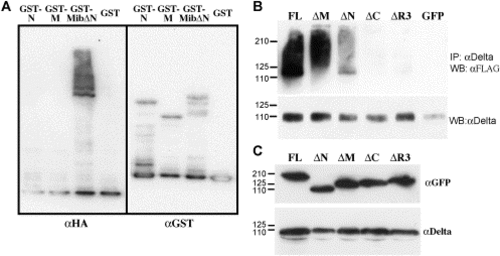
Mib is an E3 ubiquitin ligase in vitro and enhances ubiquitination of DeltaD in Cos7 cells. (A) Western blot analysis of samples from in vitro ubiquitination assays with purified GST fusion proteins of the N-terminal half, the ankyrin repeats, or the C-terminal half containing the ankyrin repeats and ring fingers shows the self-ubiquitination activity of the ring fingers in Mib. The left half is probed with anti-HA antibodies to label ubiquitin, while the right half is probed with anti-GST antibody after stripping to label the fusion proteins. (B) Western blot analysis of the ubiquitination status of DeltaD after immunoprecipitating lysates from cells co-transfected with full-length and deletion mutants of Mib demonstrates that the ubiquitin ligase activity of Mib enhances deltaD ubiquitination. Upper panel probed with anti-FLAG to reveal ubiquitin. Lower panel was probed with anti-Delta after striping to show that IP worked. (C) Western blot analysis shows similar steady state levels of the expressed protein after co-transfection. |
|
Mib-Delta interaction alters their subcellular distribution. The immunofluorescent images in each row show the subcellular localization of Mib or its deletion mutants in the presence or absence of DeltaD. The first column from the left shows the subcellular localization of Mib and its deletion mutants. The second column shows the subcellular localization of DeltaD. Third column shows the merged images of the first and second columns plus nuclear stain (blue). The rightmost column shows the merged images of Mib and its deletion mutants (green) in the absence of DeltaD. (A–C) Distribution of DeltaD and EGFP when co-expressed. (D–F) Co-localization of full-length Mib and DeltaD. (H–J) MibΔN and DeltaD do not co-localize. (L–N) MibΔM co-localizes with, but fails to enhance the internalization of DeltaD. (P–R) MibΔC co-localizes with DeltaD, and is distributed on the membrane and in perinuclear regions. (T) Distribution of full-length Mib partially overlaps that of Texas-Red-conjugated Dextran. (U) Full-length Mib, when expressed in zebrafish embryos after synthetic RNA injection, is also distributed in a punctuate manner. |
Reprinted from Developmental Biology, 267(2), Chen, W., and Casey Corliss, D., Three modules of zebrafish Mind bomb work cooperatively to promote Delta ubiquitination and endocytosis, 361-373, Copyright (2004) with permission from Elsevier. Full text @ Dev. Biol.