- Title
-
Wnt2bb Induces Cardiomyocyte Proliferation in Zebrafish Hearts via the jnk1/c-jun/creb1 Pathway
- Authors
- Peng, X., Fan, S., Tan, J., Zeng, Z., Su, M., Zhang, Y., Yang, M., Xia, L., Fan, X., Cai, W., Tang, W.H.
- Source
- Full text @ Front Cell Dev Biol
|
Induction of |
|
|
|
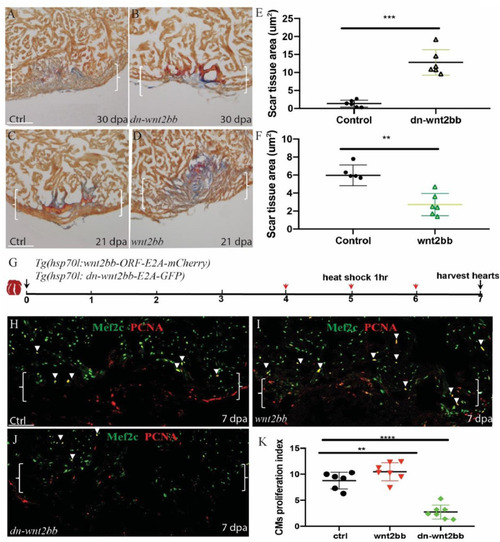
Regulation of injury-induced cardiomyocyte proliferation via |
|
|
|
|
|
Non-canonical and canonical Wnt signaling antagonize each other to regulate cardiomyocyte proliferation during zebrafish heart regeneration. |