- Title
-
Cellular rescue in a zebrafish model of congenital muscular dystrophy type 1A
- Authors
- Hall, T.E., Wood, A.J., Ehrlich, O., Li, M., Sonntag, C.S., Cole, N.J., Huttner, I.G., Sztal, T.E., Currie, P.D.
- Source
- Full text @ NPJ Regen Med
|
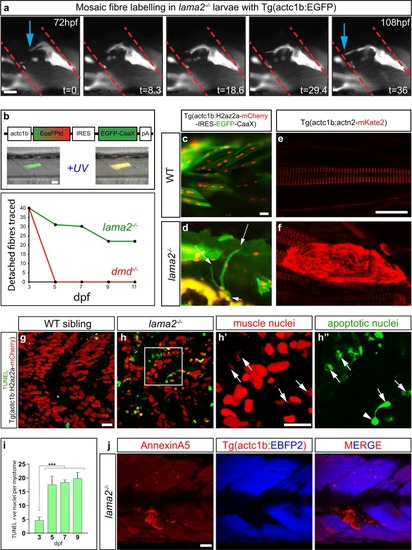
Dystrophic fibres in |
|
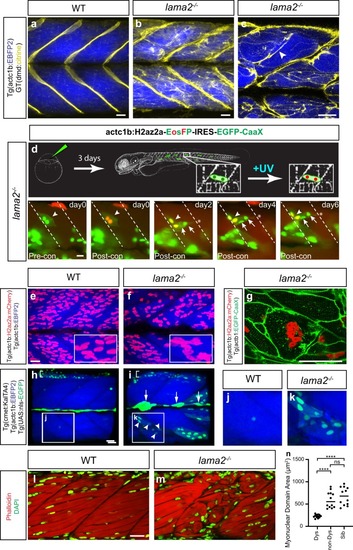
Long-lived dystrophic |
|
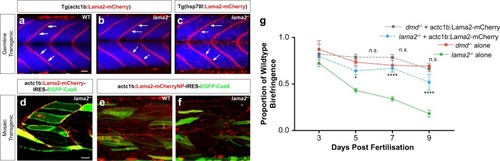
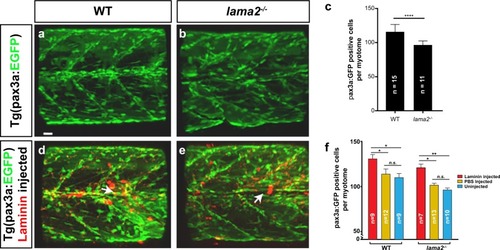
Transgenic delivery of laminin alleviates dystrophic pathology in |
|
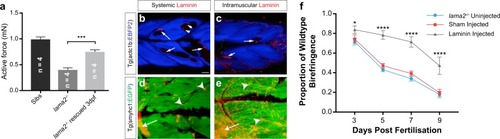
Laminin can rescue pathology in PHENOTYPE:
|
|
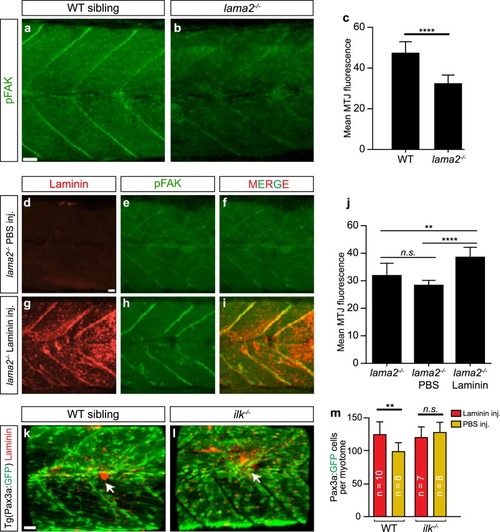
PHENOTYPE:
|
|
Laminin stimulation of stem cell proliferation requires downstream integrin signalling. |