- Title
-
Diabetic pdx1-mutant zebrafish show conserved responses to nutrient overload and anti-glycemic treatment
- Authors
- Kimmel, R.A., Dobler, S., Schmitner, N., Walsen, T., Freudenblum, J., Meyer, D.
- Source
- Full text @ Sci. Rep.
|
Pdx1sa280 encodes the nonsense mutation Y37X. (a) Schematic of the zebrafish Pdx1 protein and sequence alignment showing the location of amino acid change Y37X resulting from mutant allele pdx1sa280. The mutated tyrosine at position 37, located within the highly conserved N-terminal transactivation domain of pdx1, causes a premature stop codon. (b) Sequencing of genomic DNA from 6-month-old adult offspring of a heterozygous incross revealed surviving homozygotes. (c) Genotype frequency at 6 months of incrossed heterozygotes. Surviving homozygous mutant pdx1/ fish were present at a lower than expected frequency. (Combined data from 4 independent crosses) (d) At 3 months of age, adult mutants (right) had normal morphology but reduced size compared to wild type controls (left). Scale bars, 0.5cm. (e) Homozygous mutants had decreased length, weight and BMI as compared to wild types. n(pdx1+/+) = 18, n(pdx1/) = 19, ***p < 0.0001 (t-test). PHENOTYPE:
|
|
Pancreatic histology of pdx1 mutants. (a–d) Gut region morphology in longitudinal-oblique sections of 12 dpf wild type (a,c), and pdx1 mutant (b,d) larvae stained with hemotoxylin and eosin (H&E). (a–d) Pancreatic tissue is located between the intestinal bulb (ib) and gall bladder (gb). (li, liver) (c) In wild type embryos, exocrine cells (exo) adjacent to the islet (is) show clear polarity and acinar organization (yellow outline). Inset, close-up of single acinar cell, showing basophilic basal region (ba) and eosinophilic apical region (ap), where secretory granules are located. (d) In pdx1 mutants cell polarity and acini are not apparent. In brightfield images of an adjacent region, exocrine secretory granules can be discerned in the wild type pancreas (e, arrows) in the region immunostained for CPA (green) (g). (f,h) CPA-positive exocrine tissue in the pdx1 mutant appears disorganised. Beta cells of the islet are labeled by immunostaining for Ins (red). Nuclei are counterstained with DAPI (blue). ((a,b), scale bar = 50µm; (c–f), scale bar = 20µm) In adults, Ins + cells (red) occupy the core of the islet, while Gcga+ cells (cyan) are found mostly at the periphery in both controls and pdx1 mutants (i–l). Scale bar=50µm. (m) Polarized exocrine cells (exo) arranged in acini (yellow outline) are found adjacent to the islet in controls Inset, close-up of a single acinar cell, showing basally (ba) located nucleus and eosinophilic apical region (ap), containing secretory granules. (n) In pdx1 mutants, exocrine cells appear unpolarized and lack acinar organization. ((m,n) scale bar = 20µm) |
|
Islet formation but impaired expansion in pdx1 mutants. (a) Immunostaining for insulin (Ins, magenta) and GFP protein (green) in wild type and mutant NeuroD:eGFP+ embryos at 3 dpf. Maximum intensity projection (MIP) of confocal stacks. Scale bar, 20µm. (b) Number of Ins-expressing cells from embryos as shown in (a). (wt, n = 9; mu, n = 10) **p < 0.001 (t-test). The line indicates the median, boxes span the 25th to 75th percentile, whiskers the 10th to 90th percentile. (dpf, days post fertilization) (c, top) Schematic of experiment to evaluate feeding induction of secondary islets. Embryos were provided with nutrient powder (SDS100) from day 5 until day 7, and imaged live on day 8 to detect secondary islet cells. (c, bottom) NeuroD:eGFP+ endocrine cells are detected outside of the islet in control pdx1+/+ larvae (left), which are absent in pdx1-/- mutants (right). The pancreas is outlined (gray) based on a simultaneously acquired brightfield image. (d) Quantitation of secondary islets formed in wild type (n = 11) and mutant (n = 11) larvae treated as in (c), ***p < 0.0001, t-test. (e) Representative 7 dpf NeuroD:eGFP+ larvae treated with Ly411575 starting on day 4 and examined live by fluorescence microscopy. Heterozygous and control larvae, but not mutants, show robust induction of new GFP+ endocrine cells in the pancreatic tail (arrows). (f) Quantitation of second wave islet cells from NeuroD:eGFP+ larvae treated as in (e). ***p < 0.0001; **p < 0.001; n.s., not significant (one-way ANOVA, Tukey Multiple Comparison test). n(pdx1+/+) = 8, n(pdx1+/-) = 13, n(pdx1-/-) = 7. In (c) and (e), the fluorescent signal in the prinicipal islet has been overexposed to reveal single cells and clusters in the surrounding pancreas. Single cells and clusters were quantitated as secondary islets. EXPRESSION / LABELING:
PHENOTYPE:
|
|
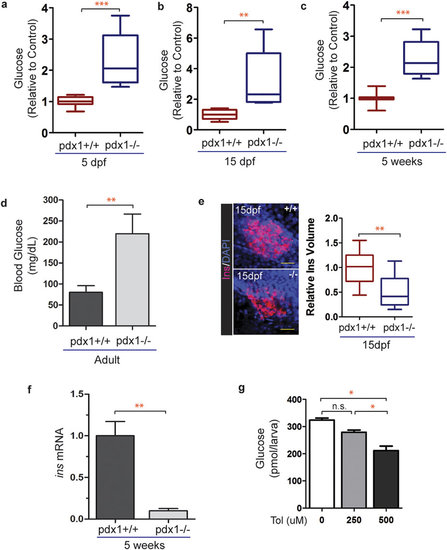
Glucose homeostasis is disrupted in pdx1 mutants. (a) Quantitation of glucose levels from 5 dpf whole larval extracts (pools of 10 embryos). n = 4 biological replicates, results combined from 3 independent experiments (***p < 0.0001, t-test). (b) Glucose levels measured at 15 dpf in extracts from pools of 3 fish, normalized to the average control value. n = 4 biological replicates, results are combined from 3 independent experiments (**p < 0.01, t-test). (c) Glucose levels relative to control from 5-week single fish (wild type, n = 7; mutant, n = 10, ***p < 0.0001, t-test). In box plots, line shows the median, box extends from the 25th to the 75th percentile, whiskers indicate 10th and 90th percentiles. (d) Adult blood glucose levels. Values are mean ± SEM. (pdx1+/+, n = 13; pdx1-/-, n = 9; **p < 0.01; t-test.) (e) Pancreas of wild type and mutant zebrafish at 15 dpf, immunostained with insulin (Ins) antibody, nuclei are counterstained with DAPI (left). Scale bar, 20µm. Quantitation of the volume of insulin staining (right). Results combined from 2 independent experiments. (pdx1+/+, n = 15; pdx1-/-, n = 16) **p < 0.001 (t-test). (f) Relative insulin mRNA levels as determined by qPCR in 5-week old wild type and pdx1-/- mutant fish. **p < 0.01 (t-test) (g) Treatment of pdx1 mutants at 5 dpf with 500 uM tolbutamide for 2 hours lowered glucose levels by 36%. *p < 0.05 (one-way ANOVA, Tukey’s Multiple Comparison Test). Results shown are representative of 2 independent experiments. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Feeding effects on beta cell induction and viability in pdx1 mutants. (a) Timeline schematic of larval feeding and analyses. (b) MIPs of confocal images of the principal islet from NeuroD:eGFP+ control (top) and pdx1 mutant (bottom) larvae fed HFD from 5–7 dpf, and analyzed at 8 dpf by TUNEL labeling and immunostaining for GFP. Nuclei were counterstained with DAPI (blue). Arrows indicate eGFP+/TUNEL+ islet cells. Scale bar, 20µm. (c) Quantitation of the percentage of apoptotic cells (TUNEL+/GFP+) in the principal islet of wild type and mutant larvae as shown in (b). Line indicates median. *p < 0.05, Mann-Whitney, one-tailed t-test. (d) Representative MIP confocal images of islets from 12 dpf Neurod:eGFP+ wild type and mutant larvae fed powder (MIN) or high fat diet (HFD) from 5–11 dpf, followed by immunostaining for GFP and Ins (magenta). Nuclei were counterstained with DAPI (blue). Scale bar, 20µm. (e) Number of NeuroD:eGFP+ islet cells in 12 dpf larvae treated as in (d). For pdx1+/+ and pdx1-/-, comparing MIN to HFD treated, *p < 0.05, Mann-Whitney, two-tailed t-test. (f) Number of Ins-expressing cells from mutant larvae as in (d) (**p < 0.01; t-test). In box plots, the median is indicated, boxes span the 25th to 75th percentile, whiskers the 10th to 90th percentile. In all graphs, number of larvae analyzed per group (combined from 2 or more independent experiments) as shown. |