- Title
-
Genetic lineage labeling in zebrafish uncovers novel neural crest contributions to the head, including gill pillar cells
- Authors
- Mongera, A., Singh, A.P., Levesque, M.P., Chen, Y.Y., Konstantinidis, P., and Nüsslein-Volhard, C.
- Source
- Full text @ Development
|
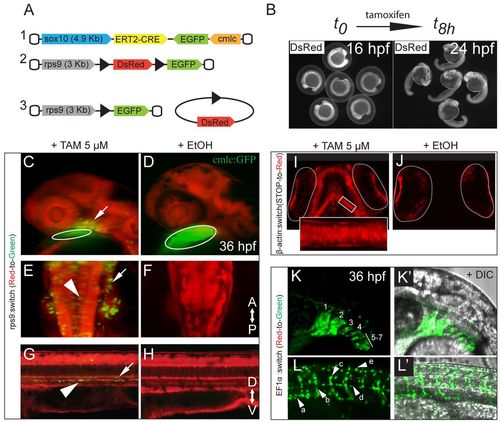
An inducible Cre/loxP-based genetic system for long-term fate mapping of zebrafish NC. (A) Schematic of the two constructs used to generate (1) a NC-specific ERT2-Cre driver line [(–4.9kb)sox10 zebrafish promoter] and (2) a ubiquitous reporter line [(–3kb)rps9 promoter from Takifugu rubripes] that enables detection of the recombination events during the early phases of NC differentiation. (3) DsRed excision after Cre-mediated recombination. (B) Time window for tamoxifen induction (16-24 hpf). (C-H) Tg(sox10:ERT2-Cre;rps9:switch) embryos at 36 hpf, treated with tamoxifen (C,E,G) or with ethanol as control (D,F,H). (C-L′) Maximum intensity projections (MIPs) of confocal stacks. (C) GFP+ recombined cells in the branchial arch region (arrow) and cmlc:GFP marker (circled). (E) Hindbrain area with recombined cells in the otic epithelium (arrow) and along the dorsal neural tube, characterized by an epithelial, pre-delamination shape (arrowhead). (G) Sagittal section of the trunk with GFP+ melanoblasts and sympathetic neuron precursors flattened along the dorsal aorta walls (arrowhead) and the ventral notochord (arrow). (D,F,H) In the uninduced controls, no recombined cells are detectable. (I,J) Developing viscerocranium in 4-dpf Tg(sox10:ERT2-Cre;β-actin:switch) larvae showing labeled chondrocytes that are absent in the control. The eyes, which show red autofluorescence, are circled. (K-L′) Tg(sox10:ERT2-Cre;EF1α:switch) induced embryos (36 hpf) showing (K,K′) labeled pharyngeal arches (1-7) and (L,L′) pigment precursors (a), motor axon glia (b), dorsal root ganglia (DRG) progenitors (c), glia along the lateral line (d) and dorsal melanoblasts (e). The anteroposterior (AP) and dorsoventral (DV) axes are indicated. EXPRESSION / LABELING:
|
|
Detection of postcranial NC derivatives. (A-D′) Lateral perspectives of epifluorescent acquisitions of DsRed+ components of the peripheral nervous system in induced Tg(sox10:ERT2-Cre;β-actin:switch) metamorphic fish. (A) Sympathetic ganglia (white) located along the ventral walls of the dorsal aorta (the dorsal walls are highlighted by the dashed line) and axonal projections (arrowheads) towards the spinal chord. (B) Neurons located in the most posterior region of the gut (dashed lines), close to the anus, forming the enteric nervous system. (C) DRG with afferent (arrow) and efferent (arrowhead) projections. (D,D′) Somatosensory projections innervating the pectoral fin (D) and the caudal fin (D′); indicated are two of the most posterior DRG in the caudal region (arrowheads) and their projections entering the fin region (arrows). (E-F′) DsRed+ glia cells in induced Tg(sox10:ERT2-Cre;β-actin:switch) metamorphic and adult fish. MIP of confocal stacks showing glia cells along the lateral line nerve (arrowhead) and the axonal projections innervating the neuromasts (arrows) in metamorphic (E) and adult (F) zebrafish. (F′) Lateral line glia in the caudal fin. (F,F′) Epifluorescence pictures. (G-I′) MIP of confocal stacks showing pigment cells in induced Tg(sox10:ERT2-Cre;β-actin:switch) metamorphic fish: (G,G′) iridophore clone, (H,H′) melanophore clone with glia cells (arrowheads) and (I,I′) xanthophore clone with glia cells (arrowhead). (J) Confocal image of transverse cryosection and anti-DsRed antibody staining showing a labeled melanophore (arrow) close to labeled glia cells wrapped around the DRG. (K,K′) Heart region of induced metamorphic fish carrying the ubi:switch transgene as a reporter. (K) 3D view of confocal stacks showing cmlc:GFP+ cardiomyocytes (gray) and mCherry+ cells (red, arrow and arrowhead) in the heart. (K′) Confocal section of the region indicated in K by the arrow, showing colocalization between an mCherry+ cell and a GFP+ cardiomyocyte. (L) Confocal image of transverse cryosection and anti-DsRed antibody staining showing an induced Tg(sox10:ERT2-Cre;β-actin:switch) metamorphic fish. Labeled adrenomedullary cells in the liver (red, inset). The gut is indicated by the asterisk. (M,M′) Confocal image of sagittal cryosection and anti-mCherry antibody staining showing the pronephric duct region of an induced Tg(sox10:ERT2-Cre;ubi:switch) metamorphic fish. (M) mCherry+ cells populate the epithelial walls of the pronephric duct. (M′) The arrow indicates the otic capsule in the posterior chondrocranium. |
|
NC-derived elements in the developing chondrocranium and viscerocranium. (A) Confocal section of recombined tissue in the ethmoid plate (e) and the trabeculae (t) of a 5-dpf Tg(sox10:ERT2-Cre;ubi:switch) larva. The red dashed lines highlight the autofluorescence of the eyes. (B) Epifluorescent image of labeled elements of the dorsal neurocranium in 15-dpf Tg(sox10:ERT2-Cre;β-actin:switch) fish: taenia marginalis anterior (tma), taenia marginalis posterior (tmp) and epiphyseal bar (eb). (C-E) MIP of confocal stacks. (C) Recombined cells in the forming ventral cartilage of the otic capsule (oc, arrowheads) in 5-dpf Tg(sox10:ERT2-Cre;ubi:switch) larvae. (D) Labeled viscerocranial elements in the mandibular region [Meckel′s cartilage (m), palatoquadrate (pq)] and in the hyod region [hyosymplectic (hs), ceratohyal (ch), basihyal (bh)] of 5-dpf Tg(sox10:ERT2-Cre;ubi:switch) larvae. (E) Labeled basibranchial (bb), hypobranchial (hb) and ceratobranchial (cb) elements of 5-dpf Tg(sox10:ERT2-Cre;ubi:switch) larvae. |
|
Detection of contested cranial NC derivatives. (A-A′′) MIP of confocal stacks showing labeled cartilaginous and bony elements in the otic capsule: chondroblasts and osteoblasts in the ventral cartilage (A′) and in the inner ear structures (A′′). (B) MIP of confocal stacks (ventral view) showing labeled viscerocranial structures: (1) Meckel′s cartilage (paired), (2) the basihyal (unpaired), (3) the ceratohyal (paired), (4) the basibranchial (unpaired) and (5) the branchiostegal rays (paired), with individual rays marked with an asterisk. (B′) Confocal section from B showing labeled (2) basihyal (unpaired), (3) ceratohyal (paired) and (4) basibranchial (unpaired) elements. (C,D) Epifluorescent image of mCherry+ osteoblasts forming the opercular series in Tg(sox10:ERT2-Cre;ubi:switch) adult fish (C) and Alizarin Red-stained skull (lateral view) in a 3-month-old zebrafish showing the four bones forming the opercular series: (1) preopercle, (2) interopercle, (3) subopercle and (4) opercle (inset). (D) 3D view of confocal stacks (boxed region in C), which shows positive cells in all the bones composing the opercular series. (E) Frontal (Fr) and parietal (P) bones. Epifluorescence image showing recombined osteoblasts in the rostral part of the frontals (arrowhead), whereas the parietals are devoid of labeled osteoblasts (arrow indicates a pigment cell clone). (F) MIP of confocal stacks showing labeled osteoblasts in the anterior region of the frontals. (G) MIP of confocal stacks of a maxillary barbel in induced adult fish showing recombined tissues in the core of the tactile organ. (H) Confocal section showing, in the red channel, labeled nerves (asterisks), fibroblast-like cells in the rod matrix (arrowheads) and flattened cells along the dorsal aspect of the rod (arrow). In the green channel, a non-switched lymphatic vessel (arrow) and blood vessel (arrowhead) are indicated. In the merge panel, the different layers composing the barbels are shown. |
|
NC origin of gill pillar cells. (A-E) Recombined cells in the primary and secondary lamellae of the gills. (A) 3D view of confocal stacks showing the gill lamellae in an induced Tg(sox10:ERT2-Cre;ubi:switch) adult fish: non-NC-derived tissues are in gray (GFP), whereas NC-derived tissues in the primary (arrow) and in the secondary (arrowhead) lamellae are in red (mCherry). (B) Epifluorescence image of DsRed+ cells in the primary and secondary lamellae in an induced Tg(sox10:ERT2-Cre;β-actin:switch) adult fish. (C) Confocal section of labeled smooth muscles of the blood vessel tunica media in the primary lamellae (arrows) and pillar cell in contact with the smooth muscle layer (arrowhead). (C2) flk:GFP+ endothelium in the gills surrounded by NC-derived smooth muscles. (D) Confocal section of a secondary lamellae showing recombined mCherry+ pillar cells surrounded by non-recombined GFP+ epithelial cells in an induced Tg(sox10:ERT2-Cre;ubi:switch) adult fish. The arrow indicates the pillar cell body, whereas the arrowhead marks a cytoplasmic process connecting two neighboring pillar cells. (E) MIP of confocal stacks showing a phalloidin-stained (gray) secondary lamella with the GFP+ pillar cell nuclei of an induced hs:R to nG adult fish. The pillar cell nuclei (arrow) are located between adjacent lumina (arrowheads). (F) Schematic of gill lamellae and pillar cells demonstrating the flow of blood through the organ. (G) MIP of confocal stacks of recombined lamellae using the crestin promoter, showing labeled smooth muscles (arrow) and pillar cells (inset). |
|
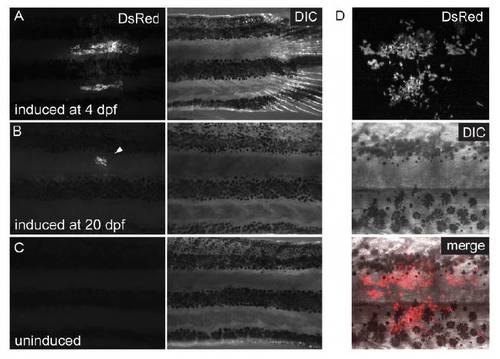
Control of postembryonic Cre induction. (A-C) Epifluorescent acquisitions and brightfield images. (A) Pigment cell clones in adult fish induced at 4 dpf and (B) 20 dpf (arrowhead). (C) Trunk of uninduced adult fish devoid of clones. (D) MIP of confocal stacks showing labeled melanophores and iridophores on the adult fish flank. |
|
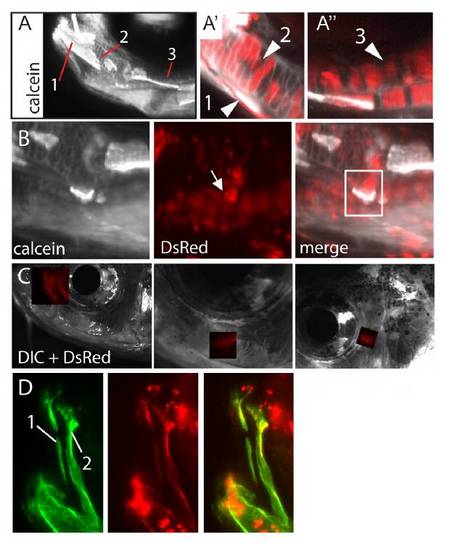
Examples of NC-derived structures detectable in vivo in metamorphic and adult fish. (A-D) Induced Tg(sox10:ERT2-Cre;β-actin:switch) metamorphic and adult fish stained with calcein. (A-A′′′) 20-dpf fish with labeled dentary (A′, 1), Meckel’s cartilage (A′, 2) and palatoquadrate (A′′, 3). (B) 20-dpf fish with labeled retroarticular bone (arrow). (C) Labeled circumorbital bones. (D) Labeled pre-maxilla (1) and maxilla (2). |
|
Recombined tissues in barbels of adult Tg(crestin:gal4-UAS:GFP;β-actin:switch) fish injected at the 1-cell stage with UAS:Cre DNA. |