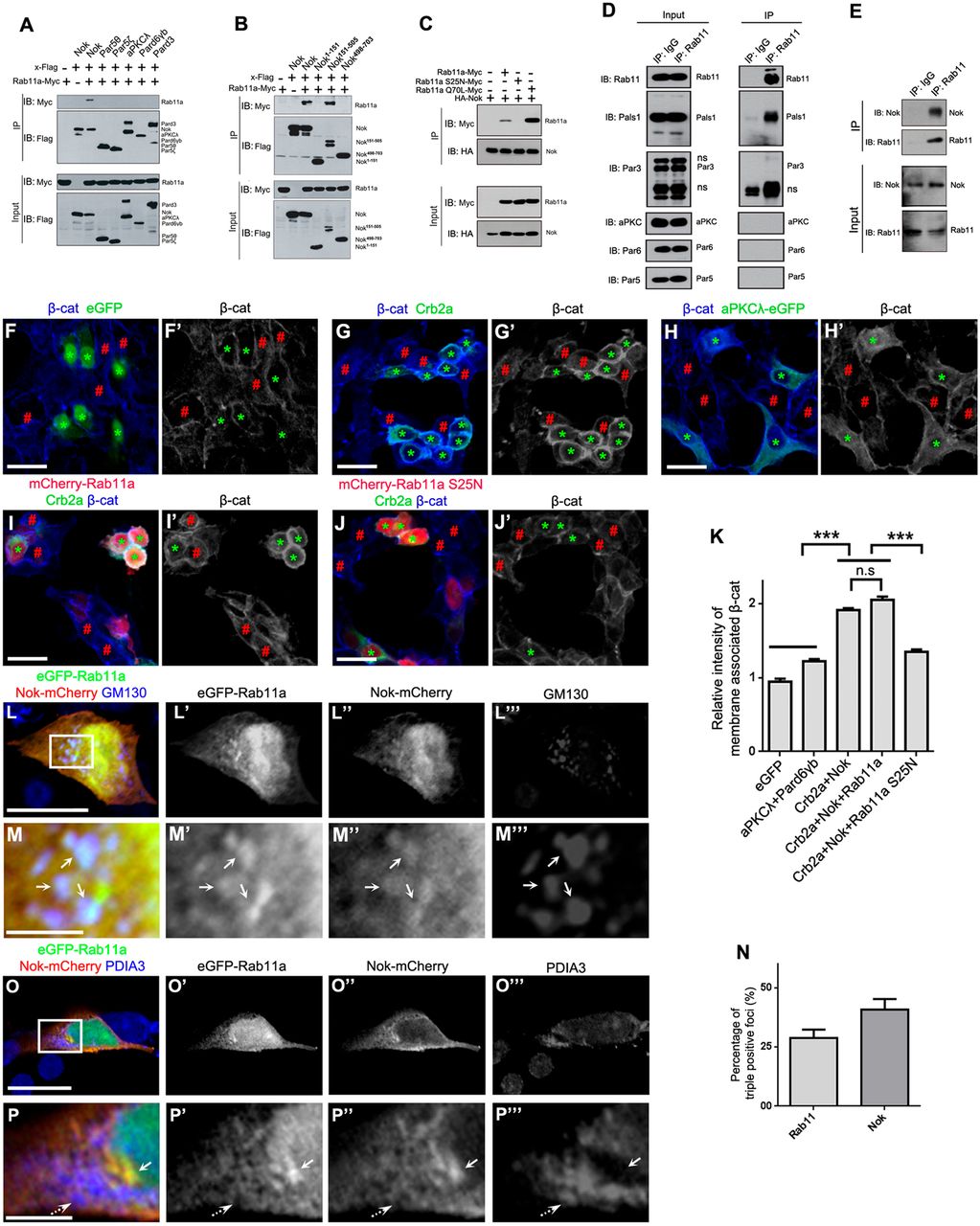
Fig. 4 Nok interplays with Rab11 to promote membrane association of β-catenin in human cultured cells. (A) Nok, but not the other polarity proteins examined, including Par5θ, Par5ζ, aPKCλ, Pard6γb and Pard3, interacted with Rab11a. (B) The L27-SH3-PDZ domain of Nok (Nok151-505) interacted with Rab11a. (C) The dominant negative Rab11a (S25N) did not interact with Nok, although the constitutively active Rab11a (Q70L) had a higher affinity for Nok than wild-type Rab11a. (D) Co-immunoprecipitation verified the endogenous complex between Rab11 and Nok in MDCK cells, but not the other tested proteins. (E) Co-immunoprecipitation using zebrafish eye extracts verified the endogenous complex between Nok and Rab11 in zebrafish. (F-H) Co-expression of Crb2a and Nok (G), but not co-expression of aPKCλ and Pard6γb (H) or expression of eGFP (F) promoted the membrane association of β-catenin in HEK293T cells. Green asterisks indicate transfection-positive cells and red # symbols indicate transfection-negative neighbor cells. (I,J) The expression of Rab11a S25N (J), but not Rab11a (I) impeded the membrane association of β-catenin induced by co-expression of Crb2a and Nok in HEK293T cells. (K) Quantification of fluorescence intensity of membrane-associated β-catenin shown in F-J (n>60 cells). Relative intensity was calculated by dividing the intensity in cells marked by an * by that in cells marked by a # symbol. (L-P) eGFP-Rab11a was colocalized with Nok-mCherry in Golgi (L,M; labeled by GM130), but not in endoplasmic reticulum (O,P; labeled by PDIA3) in MDCK cells. M and P are higher magnifications of the white boxed regions in L and O, respectively. Arrows show the aggregation of eGFP-Rab11a and Nok-mCherry, broken arrows show endoplasmic reticulum. (N) Quantification of the percentage of triple positive foci in Rab11a-positive foci and Nok-positive foci shown in L (n>60 cells). Scale bars: 20 μm (F-J), 10 μm (L,O), 2.5 μm (M,P). ***P<0.001, n.s>0.05.
Image
Figure Caption
Acknowledgments
This image is the copyrighted work of the attributed author or publisher, and
ZFIN has permission only to display this image to its users.
Additional permissions should be obtained from the applicable author or publisher of the image.
Full text @ Development