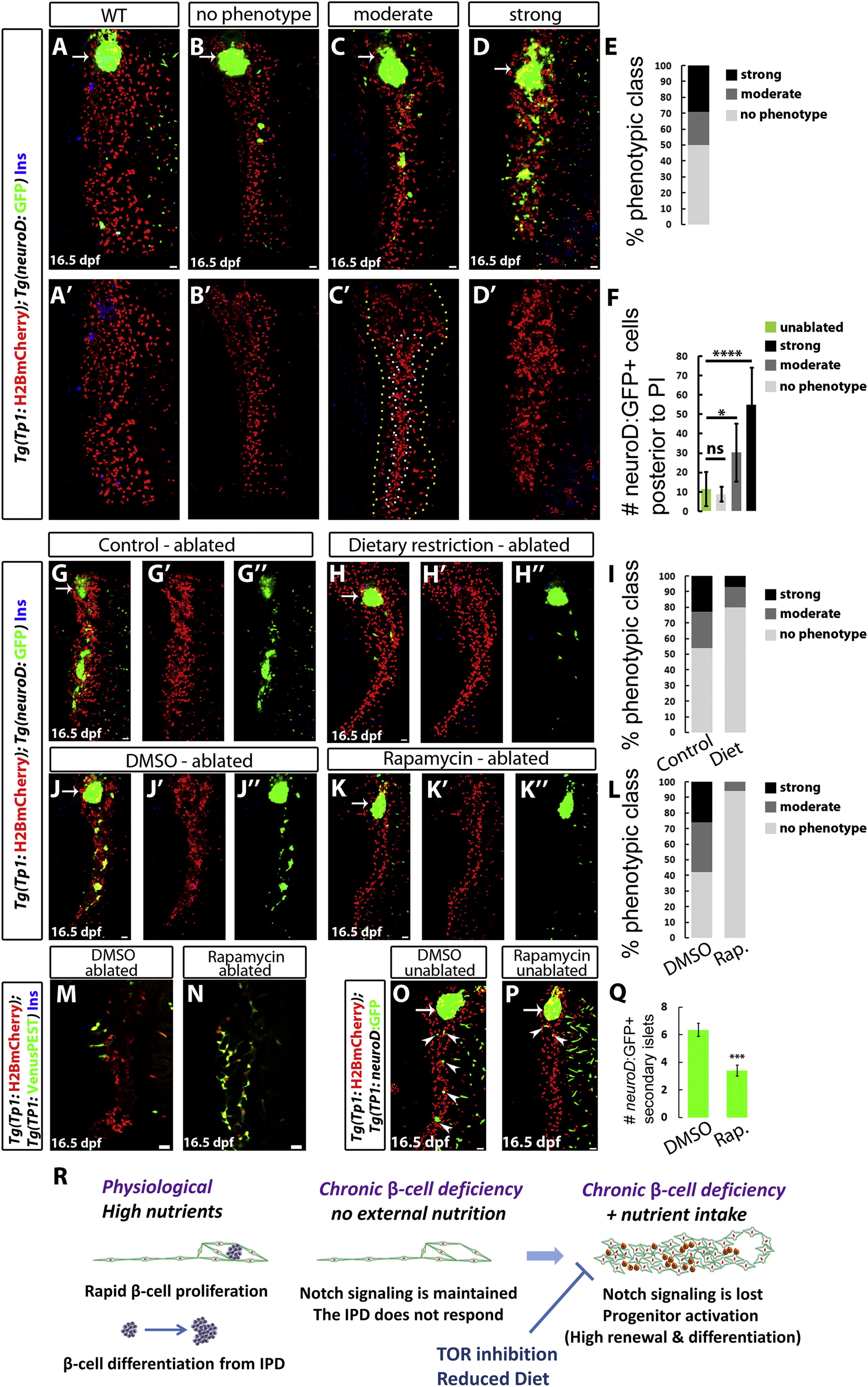
Fig. 4
High Nutrient Intake and TOR Signaling Are Required for the Activation of the Regenerative Response of IPD Cells (A–D) WT (A) and β-cell-ablated (B–D) animals at 16.5 dpf. The phenotypes of β-cell-ablated animals fall into several classes based on the degree of IPD disorganization and the extent of endocrine differentiation. (B) No phenotype; lack of IPD cell clustering and no increase in ectopic Tg(neuroD:GFP)+ cells posterior to the PI in comparison to WT animals (quantified in F). (C) Moderate phenotype; only some Tg(TP1:H2BmCherry)+ cells exhibit clustering (outlined by the white/inner dashed line in C′), whereas the rest (outlined by the yellow/outer dashed line in C′) appear unaffected. In these animals, the number of Tg(neuroD:GFP)+ cells posterior to the PI is significantly increased in comparison to WT animals (quantified in F). (D) Strong phenotype; complete clustering of the pancreatic NRCs and dramatic increase in Tg(neuroD:GFP)+ cells (quantified in F). (E) Distribution of phenotypic classes (no phenotype, 17 animals; moderate phenotype, 7 animals; and strong phenotype, 10 animals). (F) Quantification of the number of Tg(neuroD:GFP)+ cells posterior to the PI for each phenotypic class (ns, not significant; p < 0.05, ****p < 0.0001). (G and H) β-cell-ablated animals reared under a normal feeding regimen (G) or a 24 hr feeding-fasting regimen (H) (6–16 dpf). (I) Distribution of phenotypic classes for control (n = 22) versus dietary restriction (n = 15). The restricted diet suppressed the strong and moderate IPD phenotypes. (J and K) β-cell-ablated animals treated with DMSO (J) or 50 nM rapamycin (K) (8–16 dpf). The DMSO-treated animals exhibit clustering of the pancreatic NRCs as well as numerous Tg(neuroD:GFP)+ cells in the pancreatic tail, whereas, in rapamycin-treated animals, the NRCs did not undergo clustering or endocrine differentiation. (L) Distribution of phenotypic classes for animals treated with DMSO (n = 31) versus rapamycin (n = 16). Rapamycin treatment strongly suppressed the phenotypes observed in β-cell-ablated animals. (M and N) Tg(TP1:H2BmCherry); Tg(TP1:VenusPEST) β-cell-ablated animals treated with DMSO (M) or 50 nM rapamycin (N) (6–16 dpf). Of the ten DMSO-treated animals, three exhibited a loss of Tg(TP1:VenusPEST) expression in the IPD, whereas, in the rapamycin-treated animals, Tg(TP1:VenusPEST) expression was maintained (n = 15 animals). (O and P) WT animals treated with DMSO (O) or 50 nM rapamycin (P) (6–16 dpf). The DMSO-treated animal exhibits five Tg(neuroD:GFP)+ SIs (arrowheads) in its pancreatic tail, whereas the rapamycin-treated one exhibits two SIs (arrowheads). (Q) Quantification of the number of SIs posterior to the PI for DMSO- and rapamycin-treated WT animals (n = 28 for DMSO; 27 for rapamycin). Rapamycin reduced the formation of new SIs (***p < 0.001). Error bars in (F) = SD, error bars in (Q) = SEM. (R) Under physiological conditions, high nutrient intake induces β cell proliferation as well as a progressive differentiation of new β cells from the IPD. When the differentiated β cells are ablated, the combination of β cell deficiency and nutrient intake leads to the downregulation of Notch signaling in the IPD. The IPD cells undergo differentiation toward the endocrine lineages as well as an increase in proliferation and loss of ductal characteristics. This process is nutrient dependent, given that it requires high nutrient intake and can be suppressed by a dietary restriction or TOR signaling downregulation. All panels show lateral views, anterior to the top, dorsal to the left. (M and N) show single planes through the IPD, and all other panels show projections of stacks. Scale bars represent 20 μm. See also Figure S4.